Abstract
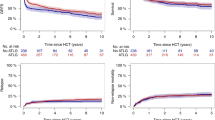
Allogeneic hematopoietic cell transplantation (allo-HCT) remains the only curative treatment option for myelofibrosis (MF) despite the emergence of novel targeted therapies. To reduce graft rejection and graft-versus-host disease (GvHD), current allo-HCT protocols often include in vivo T lymphocyte depletion using polyclonal anti-thymocyte globulin (ATG). Shortly after ATG administration, an immediate inflammatory response with fever, chills, and laboratory alterations such as cytopenias, elevation of serum C-reactive protein, bilirubin, and transaminases can develop. Here, we explore whether MF patients, who commonly exhibit extramedullary hematopoiesis in the liver, might be particularly susceptible to ATG-induced liver toxicity. To test this hypothesis, we analyzed 130 control and 94 MF patients from three transplant centers treated with or without ATG during the allo-HCT conditioning regimen. Indeed, hyperbilirubinemia was found in nearly every MF patient treated with ATG (MF-ATG 54/60 = 90 %) as compared to non-ATG treated MF (MF-noATG 15/34 = 44.1 %, p < 0.001) and respectively ATG-treated non-MF patients of the control group (control-ATG, 43/77 = 56 %, p < 0.001). In contrast, transaminases were only inconsistently elevated. Hyperbilirubinemia was in most cases self-limiting and not predictive of increased incidence of non-relapse mortality, hepatic sinusoidal obstruction syndrome (SOS) or liver GvHD. In sum, awareness of this stereotypic bilirubin elevation in MF patients treated with ATG provides a relatively benign explanation for hyperbilirubinemia occurring in these patients during the early transplant. However, attention to drug levels of biliary excreted drugs is warranted, since altered bile flow may influence their clearance and enhance toxicity (e.g., busulfan, antifungal agents).


Similar content being viewed by others
References
Kröger N, Holler E, Kobbe G, Bornhäuser M, Schwerdtfeger R, Baurmann H, Nagler A, Bethge W, Stelljes M, Uharek L (2009) Allogeneic stem cell transplantation after reduced-intensity conditioning in patients with myelofibrosis: a prospective, multicenter study of the chronic leukemia working party of the European group for blood and marrow transplantation. Blood 114(26):5264–5270
Kröger N, Deeg J, Olavarria E, Niederwieser D, Bacigalupo A, Barbui T, Rambaldi A, Mesa R, Tefferi A, Griesshammer M (2015) Indication and management of allogeneic stem cell transplantation in primary myelofibrosis: a consensus process by an EBMT/ELN international working group., Leukemia
Bonifazi F, Solano C, Wolschke C, Patriarca F, Pini M, Nagler A, Selleri M, Messina G, Bethge WA, Fuente PH (2014) Prevention of chronic GvHD after HLA-identical sibling peripheral hematopietic stem cell transplantation with or without anti-lymphocyte globulin (ATG). results from a prospective, multicenter randomized phase III trial (ATGfamilystudy). Blood 124(21):37–37
Finke J, Bethge WA, Schmoor C, Ottinger HD, Stelljes M, Zander AR, Volin L, Ruutu T, Heim DA, Schwerdtfeger R (2009) Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: a randomised, open-label, multicentre phase 3 trial. lancet Oncol 10(9):855–864
Samuelson S, Sandmaier BM, Heslop HE, Popat U, Carrum G, Champlin RE, Storb R, Prchal JT, Gooley TA, Joachim Deeg H (2011) Allogeneic haematopoietic cell transplantation for myelofibrosis in 30 patients 60–78 years of age. Br J Haematol 153(1):76–82
Kröger N, Giorgino T, Scott BL, Ditschkowski M, Alchalby H, Cervantes F, Vannucchi A, Cazzola M, Morra E, Zabelina T (2015) Impact of allogeneic stem cell transplantation on survival of patients less than 65 years of age with primary myelofibrosis. Blood 125(21):3347–3350
Pihusch R, Holler E, Mühlbayer D, Göhring P, Stötzer O, Pihusch M, Hiller E, Kolb HJ (2002) The impact of antithymocyte globulin on short-term toxicity after allogeneic stem cell transplantation. Bone Marrow Transplant 30(6):347–354
Mohty M (2007) Mechanisms of action of antithymocyte globulin: T-cell depletion and beyond. Leukemia 21(7):1387–1394
Binkert L, Medinger M, Halter JP, Heim D, Gerull S, Holbro A, Lengerke C, Weisser M, Passweg JR (2015) Lower dose anti-thymocyte globulin for GvHD prophylaxis results in improved survival after allogeneic stem cell transplantation. Bone Marrow Transplant. doi:10.1038/bmt.2015.148
Tefferi A, Lasho TL, Begna KH, Patnaik MM, Zblewski DL, Finke CM, Laborde RR, Wassie E, Schimek L, Hanson CA, Gangat N, Wang X, Pardanani A (2015) A pilot study of the telomerase inhibitor imetelstat for myelofibrosis. N Engl J Med 373(10):908–919
Weber M, Kröger N, Langer F, Hansen A, Zabelina T, Eifrig B, Hossfeld DK, Zander AR (2003) Non-overt disseminated intravascular coagulation in patients during treatment with antithymocyte globulin for unrelated allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 31(9):817–822
Bearman SI (1995) The syndrome of hepatic veno-occlusive disease after marrow transplantation. Blood 85(11):3005–3020
Shulman H, Hinterberger W (1992) Hepatic veno-occlusive disease--liver toxicity syndrome after bone marrow transplantation. Bone Marrow Transplant 10(3):197–214
Jones RJ, Lee KS, Beschorner WE, Vogel VG, Grochow LB, Braine HG, Vogelsang GB, Sensenbrenner LL, Santos GW, Saral R (1987) Venoocclusive disease of the liver following bone marrow transplantation. Transplantation 44(6):778–783
Holtan SG, Pasquini M, Weisdorf DJ (2014) Acute graft-versus-host disease: a bench-to-bedside update. Blood 124(3):363–373
Tran M-H, Fadeyi E, Scheinberg P, Klein Harvey G (2006) Apparent hemolysis following intravenous antithymocyte globulin treatment in a patient with marrow failure and a paroxysmal nocturnal hemoglobinuria clone. Transfusion 46(7):1244–1247
Kröger N, Solano C, Wolschke C, Bandini G, Patriarca F, Pini M, Nagler A, Selleri C, Risitano A, Messina G, Bethge W, Pérez De Oteiza J, Duarte R, Carella AM, Cimminiello M, Guidi S, Finke J, Mordini N, Ferra C, Sierra J, Russo D, Petrini M, Milone G, Benedetti F, Heinzelmann M, Pastore D, Jurado M, Terruzzi E, Narni F, Völp A, Ayuk F, Ruutu T, Bonifazi F (2016) Antilymphocyte globulin for prevention of chronic graft-versus-host disease. N Engl J Med 374(1):43–53
Stübig T, Alchalby H, Ditschkowski M, Wolf D, Wulf G, Zabelina T, Wolschke C, Ayuk F, Kröger N (2014) JAK inhibition with ruxolitinib as pretreatment for allogeneic stem cell transplantation in primary or post-ET/PV myelofibrosis. Leukemia 28(8):1736–1738
Wang X, Ye F, Tripodi J, Hu CS, Qiu J, Najfeld V, Novak J, Li Y, Rampal R, Hoffman R (2014) JAK2 inhibitors do not affect stem cells present in the spleens of patients with myelofibrosis. Blood 124(19):2987–2995
Geier A, Fickert P, Trauner M (2006) Mechanisms of disease: mechanisms and clinical implications of cholestasis in sepsis. Nat Clin Pract Gastroenterol Hepatol 3(10):574–585
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Prof. Finke received speaker honoraries or research support from Novartis, Neovii, Medac, Riemser.
Rights and permissions
About this article
Cite this article
Ecsedi, M., Schmohl, J., Zeiser, R. et al. Anti-thymocyte globulin-induced hyperbilirubinemia in patients with myelofibrosis undergoing allogeneic hematopoietic cell transplantation. Ann Hematol 95, 1627–1636 (2016). https://doi.org/10.1007/s00277-016-2758-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2758-z