Abstract
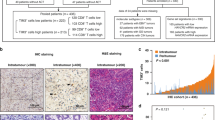
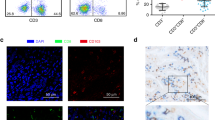
With dichotomous etiology and pathogenesis, intestinal type and diffuse type gastric cancers vary in their clinical and molecular features to the point of representing distinct entities. However, the differences of tumor-infiltrating immune cells within the two types of gastric cancer have not been well researched. This study was aimed to evaluate the functional impact of Lauren classification on immune contexture in gastric cancer patients. Tumor tissues of gastric cancer patients from Zhongshan Hospital and gastric cancer data from The Cancer Genome Atlas (TCGA) cohort were analyzed. By immunohistochemistry and flow cytometry, we found that intratumoral CD8+ T cells were more abundant but less functional in diffuse type as compared with those in intestinal type tumor tissues. Survival analysis indicated that CD8+ T cells yielded favorable prognosis only in intestinal type patients other than diffuse type cancer patients. Moreover, such diffuse type-associated CD8+ T cell dysfunction was featured by elevated expression of immunosuppressive factors including interleukin-10 (IL-10), transforming growth factor-β1 (TGF-β1) and indoleamine 2,3-dioxygenase 1 (IDO1). In summary, we found that the density, prognostic significance and functional status of intratumoral CD8+ T cells varied with Lauren subtypes in gastric cancer. These results further indicated Lauren classification might be a potential therapeutic marker, and should be considered in therapeutic decisions, especially immunotherapeutic eligibility.




Similar content being viewed by others
References
Lauren P (1965) The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. an attempt at a histological classification. Acta Pathol Microbiol Scand 64:31–49
Jimenez Fonseca P, Carmona-Bayonas A, Hernandez R et al (2017) Lauren subtypes of advanced gastric cancer influence survival and response to chemotherapy: real-world data from the AGAMENON National Cancer Registry. Br J Cancer 117:775–782. https://doi.org/10.1038/bjc.2017.245
Ma J, Shen H, Kapesa L, Zeng S (2016) Lauren classification and individualized chemotherapy in gastric cancer. Oncol Lett 11:2959–2964. https://doi.org/10.3892/ol.2016.4337
Cheng X, Yu S, Wang Y et al (2018) The role of oxaliplatin in the adjuvant setting of different Lauren's type of gastric adenocarcinoma after D2 gastrectomy: a real-world study. Gastric Cancer. https://doi.org/10.1007/s10120-018-0895-x
Network NCC (2017) (NCCN) Clinical practice guidelines in oncology. Gastric Cancer, Version 1.2017
Topalian SL, Taube JM, Anders RA, Pardoll DM (2016) Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer 16:275–287. https://doi.org/10.1038/nrc.2016.36
Wang M, Busuttil RA, Pattison S, Neeson PJ, Boussioutas A (2016) Immunological battlefield in gastric cancer and role of immunotherapies. World J Gastroenterol 22:6373–6384. https://doi.org/10.3748/wjg.v22.i28.6373
Speiser DE, Ho PC, Verdeil G (2016) Regulatory circuits of T cell function in cancer. Nat Rev Immunol 16:599–611. https://doi.org/10.1038/nri.2016.80
Sharma P, Shen Y, Wen S et al (2007) CD8 tumor-infiltrating lymphocytes are predictive of survival in muscle-invasive urothelial carcinoma. Proc Natl Acad Sci USA 104:3967–3972. https://doi.org/10.1073/pnas.0611618104
Hegde PS, Karanikas V, Evers S (2016) The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clin Cancer Res 22:1865–1874. https://doi.org/10.1158/1078-0432.CCR-15-1507
Tumeh PC, Harview CL, Yearley JH et al (2014) PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515:568–571. https://doi.org/10.1038/nature13954
Daud AI, Loo K, Pauli ML et al (2016) Tumor immune profiling predicts response to anti-PD-1 therapy in human melanoma. J Clin Investig 126:3447–3452. https://doi.org/10.1172/JCI87324
Pernot S, Terme M, Radosevic-Robin N et al (2019) Infiltrating and peripheral immune cell analysis in advanced gastric cancer according to the Lauren classification and its prognostic significance. Gastric Cancer. https://doi.org/10.1007/s10120-019-00983-3
Chen YC, Fang WL, Wang RF et al (2016) Clinicopathological variation of Lauren classification in gastric cancer. Pathol Oncol Res POR 22:197–202. https://doi.org/10.1007/s12253-015-9996-6
Huang SC, Ng KF, Yeh TS, Cheng CT, Lin JS, Liu YJ, Chuang HC, Chen TC (2019) Subtraction of Epstein–Barr virus and microsatellite instability genotypes from the Lauren histotypes: Combined molecular and histologic subty** with clinicopathological and prognostic significance validated in a cohort of 1,248 cases. Int J Cancer. https://doi.org/10.1002/ijc.32215
Lee JH, Chang KK, Yoon C, Tang LH, Strong VE, Yoon SS (2018) Lauren histologic type is the most important factor associated with pattern of recurrence following resection of gastric adenocarcinoma. Ann Surg 267:105–113. https://doi.org/10.1097/SLA.0000000000002040
Specht E, Kaemmerer D, Sanger J, Wirtz RM, Schulz S, Lupp A (2015) Comparison of immunoreactive score, HER2/neu score and H score for the immunohistochemical evaluation of somatostatin receptors in bronchopulmonary neuroendocrine neoplasms. Histopathology 67:368–377. https://doi.org/10.1111/his.12662
Davoli T, Uno H, Wooten EC, Elledge SJ (2017) Tumor aneuploidy correlates with markers of immune evasion and with reduced response to immunotherapy. Science. https://doi.org/10.1126/science.aaf8399
Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY, **ao YS, Xu Y, Li YW, Tang ZY (2007) Intratumoral balance of regulatory and cytotoxic T cells is associated with prognosis of hepatocellular carcinoma after resection. J Clin Oncol 25:2586–2593. https://doi.org/10.1200/jco.2006.09.4565
Hamanishi J, Mandai M, Iwasaki M et al (2007) Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci USA 104:3360–3365. https://doi.org/10.1073/pnas.0611533104
Donnem T, Hald SM, Paulsen EE et al (2015) Stromal CD8+ T-cell density—a promising supplement to TNM staging in non-small cell lung cancer. Clin Cancer Res 21:2635–2643. https://doi.org/10.1158/1078-0432.ccr-14-1905
Mahmoud SM, Paish EC, Powe DG, Macmillan RD, Grainge MJ, Lee AH, Ellis IO, Green AR (2011) Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer. J Clin Oncol 29:1949–1955. https://doi.org/10.1200/jco.2010.30.5037
Thompson ED, Zahurak M, Murphy A et al (2017) Patterns of PD-L1 expression and CD8 T cell infiltration in gastric adenocarcinomas and associated immune stroma. Gut 66:794–801. https://doi.org/10.1136/gutjnl-2015-310839
Wen T, Wang Z, Li Y et al (2017) A four-factor immunoscore system that predicts clinical outcome for stage II/III gastric cancer. Cancer Immunol Res 5:524–534. https://doi.org/10.1158/2326-6066.cir-16-0381
Chen DS, Mellman I (2017) Elements of cancer immunity and the cancer-immune set point. Nature 541:321–330. https://doi.org/10.1038/nature21349
Goode EL, Block MS, Kalli KR et al (2017) Dose-response association of CD8+ tumor-infiltrating lymphocytes and survival time in high-grade serous ovarian cancer. JAMA Oncol 3:e173290. https://doi.org/10.1001/jamaoncol.2017.3290
Lizotte PH, Ivanova EV, Awad MM, Jones RE, Keogh L, Liu H, Dries R, Almonte C (2016) Multiparametric profiling of non-small-cell lung cancers reveals distinct immunophenotypes. JCI Insight 1:e89014
Desmedt C, Salgado R, Fornili M et al (2018) Immune infiltration in invasive lobular breast cancer. J Natl Cancer Inst 110:768–776. https://doi.org/10.1093/jnci/djx268
Cristescu R, Lee J, Nebozhyn M et al (2015) Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med 21:449–456. https://doi.org/10.1038/nm.3850
Oh SC, Sohn BH, Cheong JH et al (2018) Clinical and genomic landscape of gastric cancer with a mesenchymal phenotype. Nat Commun 9:1777. https://doi.org/10.1038/s41467-018-04179-8
Lou Y, Diao L, Cuentas ER et al (2016) Epithelial–mesenchymal transition is associated with a distinct tumor microenvironment including elevation of inflammatory signals and multiple immune checkpoints in lung adenocarcinoma. Clin Cancer Res 22:3630–3642. https://doi.org/10.1158/1078-0432.CCR-15-1434
Mak MP, Tong P, Diao L et al (2016) A patient-derived, pan-cancer EMT signature identifies global molecular alterations and immune target enrichment following epithelial-to-mesenchymal transition. Clin Cancer Res 22:609–620. https://doi.org/10.1158/1078-0432.CCR-15-0876
Wang L, Saci A, Szabo PM et al (2018) EMT- and stroma-related gene expression and resistance to PD-1 blockade in urothelial cancer. Nat Commun 9:3503. https://doi.org/10.1038/s41467-018-05992-x
Chen L, Gibbons DL, Goswami S et al (2014) Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression. Nat Commun 5:5241. https://doi.org/10.1038/ncomms6241
Liu CY, Xu JY, Shi XY, Huang W, Ruan TY, **e P, Ding JL (2013) M2-polarized tumor-associated macrophages promoted epithelial-mesenchymal transition in pancreatic cancer cells, partially through TLR4/IL-10 signaling pathway. Lab Investig 93:844–54. https://doi.org/10.1038/labinvest.2013.69
Zhu L, Fu X, Chen X, Han X, Dong P (2017) M2 macrophages induce EMT through the TGF-beta/Smad2 signaling pathway. Cell Biol Int 41:960–968. https://doi.org/10.1002/cbin.10788
Kolijn K, Verhoef EI, Smid M, Bottcher R, Jenster GW, Debets R, van Leenders G (2018) Epithelial–mesenchymal transition in human prostate cancer demonstrates enhanced immune evasion marked by IDO1 expression. Can Res 78:4671–4679. https://doi.org/10.1158/0008-5472.CAN-17-3752
Mariathasan S, Turley SJ, Nickles D et al (2018) TGFbeta attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554:544–548. https://doi.org/10.1038/nature25501
Kang YK, Boku N, Satoh T et al (2017) Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 390:2461–2471. https://doi.org/10.1016/s0140-6736(17)31827-5
Shitara K, Ozguroglu M, Bang YJ et al (2018) Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet 392:123–133. https://doi.org/10.1016/s0140-6736(18)31257-1
Acknowledgments
Not applicable.
Funding
This study was funded by Grants from National Natural Science Foundation of China (81671628, 81672324, 31770851, 81871306, 81871926, 81871930, 81902402, 81902901, 81972219), Shanghai Municipal Natural Science Foundation (18ZR1432900) and Shanghai Sailing Program (17YF1402200, 18YF1404600, 19YF1407500). All these study sponsors have no roles in the study design, in the collection, analysis and interpretation of data.
Author information
Authors and Affiliations
Contributions
In this study, RCL, HZ, YFC and XL involved in acquisition of data, analysis and interpretation of data and drafting of the manuscript; RCL took part in the statistical analysis; YFC, YYQ, HL, JTW, KY, LLC, CL, HYH, HL, ZBS, JQ and WJZ took part in technical and material support; YHS and JJX participated in study concept and design, analysis and interpretation of data, drafting of the manuscript, obtained funding and study supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, R., Zhang, H., Cao, Y. et al. Lauren classification identifies distinct prognostic value and functional status of intratumoral CD8+ T cells in gastric cancer. Cancer Immunol Immunother 69, 1327–1336 (2020). https://doi.org/10.1007/s00262-020-02550-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02550-7