Abstract
Purpose
Hypoxia-inducible factor 2α (HIF-2α) overexpression leads to activation of angiogenic pathways. However, little is known about the association between HIF-2α expression and anti-tumor immunity in clear cell renal cell carcinoma (ccRCC). We aimed to explore how HIF-2α influenced the microenvironment and the underlying mechanisms.
Experimental design
We immunohistochemically evaluated immune cells infiltrations and prognostic value of HIF-2α expression in a retrospective Zhongshan Hospital cohort of 280 ccRCC patients. Fresh tumor samples, non-tumor tissues and autologous peripheral blood for RT-PCR, ELISA and flow cytometry analyses were collected from patients who underwent nephrectomy in Zhongshan Hospital from September 2017 to April 2018. The TCGA KIRC cohort and SATO cohort were assessed to support our findings.
Results
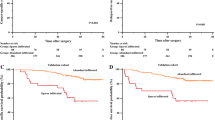
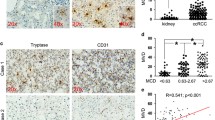
We demonstrated that ccRCC patients with HIF-2αhigh tumors exhibited reduced overall survival (p = 0.025) and recurrence-free survival (p < 0.001). Functions of CD8+ T cells were impaired in HIF-2αhigh patients. In ccRCC patients, HIF-2α induced the expression of stem cell factor (SCF), which served as chemoattractant for mast cells. Tumor infiltrating mast cells impaired anti-tumor immunity partly by secreting IL-10 and TGF-β. HIF-2α mRNA level adversely associated with immunostimulatory genes expression in KIRC and SATO cohorts.
Conclusions
HIF-2α contributed to evasion of anti-tumor immunity via SCF secretion and subsequent recruitment of mast cells in ccRCC patients.






Similar content being viewed by others
Abbreviations
- ccRCC:
-
Clear cell renal cell carcinoma
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GO:
-
Gene ontology
- GSEA:
-
Gene set enrichment analyses
- HIF:
-
Hypoxia-inducible factors
- KIRC:
-
Kidney clear cell carcinoma
- pVHL:
-
Von Hippel-Lindau (VHL) protein
- RCC:
-
Renal cell carcinoma
- SCF:
-
Stem cell factor
- TCGA:
-
The Cancer Genome Atlas
- TIM:
-
Tumor infiltrating mast cell
- VHL:
-
Von Hippel-Lindau
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917. https://doi.org/10.1002/ijc.25516
Cohen HT, McGovern FJ (2005) Renal-cell carcinoma. N Engl J Med 353:2477–2490. https://doi.org/10.1056/NEJMra043172
Gupta K, Miller JD, Li JZ, Russell MW, Charbonneau C (2008) Epidemiologic and socioeconomic burden of metastatic renal cell carcinoma (mRCC): a literature review. Cancer Treat Rev 34:193–205. https://doi.org/10.1016/j.ctrv.2007.12.001
Escudier B (2012) Emerging immunotherapies for renal cell carcinoma. Ann Oncol 23(Suppl 8):viii35–40. https://doi.org/10.1093/annonc/mds261
Atkins MB, Regan M, McDermott D (2004) Update on the role of interleukin 2 and other cytokines in the treatment of patients with stage IV renal carcinoma. Clin Cancer Res 10:6342S–6342S6S. https://doi.org/10.1158/1078-0432.CCR-040029
Nyhan MJ, O’Sullivan GC, McKenna SL (2008) Role of the VHL (von Hippel-Lindau) gene in renal cancer: a multifunctional tumour suppressor. Biochem Soc Trans 36:472–478. https://doi.org/10.1042/BST0360472
Farber LJ, Furge K, Teh BT (2012) Renal cell carcinoma deep sequencing: recent developments. Curr Oncol Rep 14:240–248. https://doi.org/10.1007/s11912-012-0230-3
Schodel J, Grampp S, Maher ER, Moch H, Ratcliffe PJ, Russo P, Mole DR (2016) Hypoxia, Hypoxia-inducible Transcription Factors, and Renal Cancer. Eur Urol 69:646–657. https://doi.org/10.1016/j.eururo.2015.08.007
Schodel J, Oikonomopoulos S, Ragoussis J, Pugh CW, Ratcliffe PJ, Mole DR (2011) High-resolution genome-wide map** of HIF-binding sites by ChIP-sEq. Blood 117:e207–e217. https://doi.org/10.1182/blood-2010-10-314427
Mole DR, Blancher C, Copley RR, Pollard PJ, Gleadle JM, Ragoussis J, Ratcliffe PJ (2009) Genome-wide association of hypoxia-inducible factor (HIF)-1alpha and HIF-2alpha DNA binding with expression profiling of hypoxia-inducible transcripts. J Biol Chem 284:16767–16775. https://doi.org/10.1074/jbc.M901790200
Raval RR, Lau KW, Tran MG et al (2005) Contrasting properties of hypoxia-inducible factor 1 (HIF-1) and HIF-2 in von Hippel-Lindau-associated renal cell carcinoma. Mol Cell Biol 25:5675–5686. https://doi.org/10.1128/MCB.25.13.5675-5686.2005
Kroeger N, Seligson DB, Signoretti S, Yu H, Magyar CE, Huang J, Belldegrun AS, Pantuck AJ (2014) Poor prognosis and advanced clinicopathological features of clear cell renal cell carcinoma (ccRCC) are associated with cytoplasmic subcellular localisation of Hypoxia inducible factor-2alpha. Eur J Cancer 50:1531–1540. https://doi.org/10.1016/j.ejca.2014.01.031
Biswas S, Charlesworth PJ, Turner GD et al (2012) CD31 angiogenesis and combined expression of HIF-1alpha and HIF-2alpha are prognostic in primary clear-cell renal cell carcinoma (CC-RCC), but HIFalpha transcriptional products are not: implications for antiangiogenic trials and HIFalpha biomarker studies in primary CC-RCC. Carcinogenesis 33:1717–1725. https://doi.org/10.1093/carcin/bgs222
Wallace EM, Rizzi JP, Han G et al (2016) A small-molecule antagonist of HIF2alpha is efficacious in preclinical models of renal cell carcinoma. Cancer Res 76:5491–5500. https://doi.org/10.1158/0008-5472.CAN-16-0473
Cho H, Kaelin WG (2016) Targeting HIF2 in clear cell renal cell carcinoma. Cold Spring Harb Symp Quant Biol 81:113–121. https://doi.org/10.1101/sqb.2016.81.030833
Palucka AK, Coussens LM (2016) The basis of oncoimmunology. Cell 164:1233–1247. https://doi.org/10.1016/j.cell.2016.01.049
Chen DS, Mellman I (2013) Oncology meets immunology: the cancer-immunity cycle. Immunity 39:1–10. https://doi.org/10.1016/j.immuni.2013.07.012
Vandyke K, Zeissig MN, Hewett DR et al (2017) HIF-2alpha promotes dissemination of plasma cells in multiple myeloma by regulating CXCL12/CXCR4 and CCR1. Cancer Res 77:5452–5463. https://doi.org/10.1158/0008-5472.CAN-17-0115
Yamamura K, Uruno T, Shiraishi A et al (2017) The transcription factor EPAS1 links DOCK8 deficiency to atopic skin inflammation via IL-31 induction. Nat Commun 8:13946. https://doi.org/10.1038/ncomms13946
Messai Y, Noman MZ, Hasmim M, Escudier B, Chouaib S (2015) HIF-2alpha/ITPR1 axis: a new saboteur of NK-mediated lysis. Oncoimmunology 4:e985951. https://doi.org/10.4161/2162402X.2014.985951
Sato Y, Yoshizato T, Shiraishi Y et al (2013) Integrated molecular analysis of clear-cell renal cell carcinoma. Nat Genet 45:860–867. https://doi.org/10.1038/ng.2699
Gentles AJ, Newman AM, Liu CL et al (2015) The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat Med 21:938–945. https://doi.org/10.1038/nm.3909
Szklarczyk D, Franceschini A, Wyder S et al (2015) STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–D452. https://doi.org/10.1093/nar/gku1003
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Benito-Martin A, Peinado H (2015) FunRich proteomics software analysis, let the fun begin! Proteomics. 15:2555–2556. https://doi.org/10.1002/pmic.201500260
Subramanian A, Tamayo P, Mootha VK et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102:15545–15550. https://doi.org/10.1073/pnas.0506580102
Wang J, Liu L, Qu Y et al (2016) Prognostic value of SETD2 expression in patients with metastatic renal cell carcinoma treated with tyrosine kinase inhibitors. J Urol 196:1363–1370. https://doi.org/10.1016/j.juro.2016.06.010
Liu H, Xu J, Zhou L, Yun X, Chen L, Wang S, Sun L, Wen Y, Gu J (2011) Hepatitis B virus large surface antigen promotes liver carcinogenesis by activating the Src/PI3K/Akt pathway. Cancer Res 71:7547–7557. https://doi.org/10.1158/0008-5472.CAN-11-2260
Wang X, Dong J, Jia L et al (2017) HIF-2-dependent expression of stem cell factor promotes metastasis in hepatocellular carcinoma. Cancer Lett 393:113–124. https://doi.org/10.1016/j.canlet.2017.01.032
Meininger CJ, Yano H, Rottapel R, Bernstein A, Zsebo KM, Zetter BR (1992) The c-kit receptor ligand functions as a mast cell chemoattractant. Blood 79:958–963
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
Disis ML (2010) Immune regulation of cancer. J Clin Oncol 28:4531–4538. https://doi.org/10.1200/JCO.2009.27.2146
Baldewijns MM, van Vlodrop IJ, Vermeulen PB, Soetekouw PM, van Engeland M, de Bruine AP (2010) VHL and HIF signalling in renal cell carcinogenesis. J Pathol 221:125–138. https://doi.org/10.1002/path.2689
Khazaie K, Blatner NR, Khan MW et al (2011) The significant role of mast cells in cancer. Cancer Metastasis Rev 30:45–60. https://doi.org/10.1007/s10555-011-9286-z
Marech I, Gadaleta CD, Ranieri G (2014) Possible prognostic and therapeutic significance of c-Kit expression, mast cell count and microvessel density in renal cell carcinoma. Int J Mol Sci 15:13060–13076. https://doi.org/10.3390/ijms150713060
Chen W, Hill H, Christie A et al (2016) Targeting renal cell carcinoma with a HIF-2 antagonist. Nature 539:112–117. https://doi.org/10.1038/nature19796
Ribatti D (2016) Mast cells as therapeutic target in cancer. Eur J Pharmacol 778:152–157. https://doi.org/10.1016/j.ejphar.2015.02.056
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331:1565–1570. https://doi.org/10.1126/science.1203486
Funding
This work was supported by grants from National Natural Science Foundation of China (81471621, 81472227, 81472376, 81671628, 31770851, 81702496, 81702497, 81702805, 81772696, 81871306), Shanghai Municipal Natural Science Foundation (17ZR1405100), Shanghai Municipal Commission of Health and Family Planning (20174Y0042), and Zhongshan Hospital Science Foundation (2016ZSQN30, 2017ZSQN18, 2017ZSYQ26). All these study sponsors have no roles in design of the study or collection, analysis, and interpretation of data.
Author information
Authors and Affiliations
Contributions
Acquisition of data, analysis and interpretation of data, statistical analysis and drafting of the manuscript were carried out by YX, LL and YX; YQ, YC, LC, PZ, YK, YQ, ZW, ZL, XC, ZX, JW, QB, WZ and YY provided technical and material support; JG and JX were responsible for the study concept and design, analysis and interpretation of data, drafting of the manuscript, obtained funding and study supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval and ethical standards
The study was approved by the Clinical Research Ethics Committee of Zhongshan Hospital, Fudan University with the approval number B2015-030. Our study followed the Helsinki declaration.
Informed consent
Informed consent to use clinical samples and information was obtained from each patient.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
**ong, Y., Liu, L., **a, Y. et al. Tumor infiltrating mast cells determine oncogenic HIF-2α-conferred immune evasion in clear cell renal cell carcinoma. Cancer Immunol Immunother 68, 731–741 (2019). https://doi.org/10.1007/s00262-019-02314-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02314-y