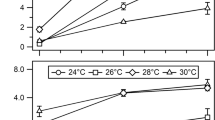
Abstract
The enzyme nitrogenase performs the process of biological nitrogen fixation (BNF), converting atmospheric dinitrogen gas into the biologically accessible ammonia, which is rapidly protonated at physiological pH to yield ammonium. The reduction of dinitrogen requires both ATP and electrons. Azotobacter vinelandii is an aerobic nitrogen-fixing microbe that is a model organism for the study of BNF. Previous reports have described strains of A. vinelandii that are partially deregulated for BNF, resulting in the release of large quantities of ammonium into the growth medium. Determining the source of the electrons required to drive BNF is complicated by the existence of several protein complexes in A. vinelandii that have been linked to BNF in other species. In this work, we used the high-ammonium-accumulating strains of A. vinelandii to probe the source of electrons to nitrogenase by disrupting the Rnf1 and Fix complexes. The results of this work demonstrate the potential of these strains to be used as a tool to investigate the contributions of other enzymes or complexes in the process of BNF. These results provide strong evidence that the Rnf1 complex of A. vinelandii is the primary source of electrons delivered to the nitrogenase enzyme in this partially deregulated strain. The Fix complex under native regulation was unable to provide sufficient electrons to accumulate extracellular ammonium in the absence of the Rnf1 complex. Increased ammonium accumulation could be attained in a strain lacking the Rnf1 complex if the genes of the Fix protein complex were relocated behind the strong promoter of the S-layer protein but still failed to achieve the levels found with just the Rnf1 complex by itself.
Key points
• The Rnf1 complex is integral to ammonium accumulation in A. vinelandii.
• The Fix complex can be deleted and still achieve ammonium accumulation in A. vinelandii.
• A. vinelandii can be engineered to increase the contribution of the Fix complex to ammonium accumulation.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article or are available from the corresponding author on reasonable request.
References
Ambrosio R, Ortiz-Marquez JCF, Curatti L (2017) Metabolic engineering of a diazotrophic bacterium improves ammonium release and biofertilization of plants and microalgae. Metab Eng 40:59–68
Bali A, Blanco G, Hill S, Kennedy C (1992) Excretion of ammonium by a nifL mutant of Azotobacter vinelandii fixing nitrogen. Appl Environ Microbiol 58(5):1711–1718
Barney BM (2020) Aerobic nitrogen-fixing bacteria for hydrogen and ammonium production: current state and perspectives. Appl Microbiol Biotechnol 104(4):1383–1399
Barney BM, Eberhart LJ, Ohlert JM, Knutson CM, Plunkett MH (2015) Gene deletions resulting in increased nitrogen release by Azotobacter vinelandii: application of a novel nitrogen biosensor. Appl Environ Microbiol 81(13):4316–4328
Barney BM, Igarashi RY, Dos Santos PC, Dean DR, Seefeldt LC (2004) Substrate interaction at an iron-sulfur face of the FeMo-cofactor during nitrogenase catalysis. J Biol Chem 279(51):53621–53624
Barney BM, Lee HI, Dos Santos PC, Hoffman BM, Dean DR, Seefeldt LC (2006) Breaking the N2 triple bond: insights into the nitrogenase mechanism. Dalton Trans (19):2277–2284
Barney BM, Lukoyanov D, Igarashi RY, Laryukhin M, Yang TC, Dean DR, Hoffman BM, Seefeldt LC (2009) Trap** an intermediate of dinitrogen (N2) reduction on nitrogenase. Biochemistry 48(38):9094–9102
Barney BM, Plunkett MH, Natarajan V, Mus F, Knutson CM, Peters JW (2017) Transcriptional analysis of an ammonium-excreting strain of Azotobacter vinelandii deregulated for nitrogen fixation. Appl Environ Microbiol 83(20):e01534-17
Batista MB, Brett P, Appia-Ayme C, Wang YP, Dixon R (2021) Disrupting hierarchical control of nitrogen fixation enables carbon-dependent regulation of ammonia excretion in soil diazotrophs. PLoS Genet 17(6):e1009617
Blanco G, Drummond M, Woodley P, Kennedy C (1993) Sequence and molecular analysis of the nifL gene of Azotobacter vinelandii. Mol Microbiol 9(4):869–879
Brewin B, Woodley P, Drummond M (1999) The basis of ammonium release in nifL mutants of Azotobacter vinelandii. J Bacteriol 181(23):7356–7362
Burén S, Jiménez-Vicente E, Echavarri-Erasun C, Rubio LM (2020) Biosynthesis of nitrogenase cofactors. Chem Rev 120(12):4921–4968
Burén S, Rubio LM (2018) State of the art in eukaryotic nitrogenase engineering. FEMS Microbiol Lett 365(2):fnx274
Burgess BK, Lowe DJ (1996) Mechanism of molybdenum nitrogenase. Chem Rev 96(7):2983–3011
Chen JG, Crooks RM, Seefeldt LC, Bren KL, Bullock RM, Darensbourg MY, Holland PL, Hoffman B, Janik MJ, Jones AK, Kanatzidis MG, King P, Lancaster KM, Lymar SV, Pfromm P, Schneider WF, Schrock RR (2018) Beyond fossil fuel-driven nitrogen transformations. Science 360(6391):eaar6611
Curatti L, Brown CS, Ludden PW, Rubio LM (2005) Genes required for rapid expression of nitrogenase activity in Azotobacter vinelandii. Proc Natl Acad Sci USA 102(18):6291–6296
Dixon R (1998) The oxygen-responsive NifL-NifA complex: a novel two-component regulatory system controlling nitrogenase synthesis in γ-proteobacteria. Arch Microbiol 169(5):371–380
Dixon RA, Postgate JR (1972) Genetic transfer of nitrogen fixation from Klebsiella pneumoniae to Escherichia coli. Nature 237(5350):102–103
Dos Santos PC (2011) Molecular biology and genetic engineering in nitrogen fixation. Methods Mol Biol 766(Nitrogen Fixation: Methods and Protocols):81–92
Earl CD, Ronson CW, Ausubel FM (1987) Genetic and structural analysis of the Rhizobium meliloti fixA, fixB, fixC, and fixX genes. J Bacteriol 169(3):1127–1136
Eberhart LJ, Knutson CM, Barney BM (2016) A methodology for markerless genetic modifications in Azotobacter vinelandii. J Appl Microbiol 120(6):1595–1604
Fellay R, Frey J, Krisch H (1987) Interposon mutagenesis of soil and water bacteria: a family of DNA fragments designed for in vitro insertional mutagenesis of Gram-negative bacteria. Gene 52(2–3):147–154
Hreha TN, Mezic KG, Herce HD, Duffy EB, Bourges A, Pryshchep S, Juarez O, Barquera B (2015) Complete topology of the RNF complex from Vibrio cholerae. Biochemistry 54(15):2443–2455
Jacobson MR, Brigle KE, Bennett LT, Setterquist RA, Wilson MS, Cash VL, Beynon J, Newton WE, Dean DR (1989) Physical and genetic map of the major nif gene cluster from Azotobacter vinelandii. J Bacteriol 171(2):1017–1027
Keen NT, Tamaki S, Kobayashi D, Trollinger D (1988) Improved broad-host-range plasmids for DNA cloning in Gram-negative bacteria. Gene 70(1):191–197
Knutson CM, Pieper MN, Barney BM (2021) Gene fitness of Azotobacter vinelandii under diazotrophic growth. J Bacteriol 203(24):e0040421
Knutson CM, Plunkett MH, Liming RA, Barney BM (2018) Efforts toward optimization of aerobic biohydrogen reveal details of secondary regulation of biological nitrogen fixation by nitrogenous compounds in Azotobacter vinelandii. Appl Microbiol Biotechnol 102(23):10315–10325
Ledbetter R, Garcia-Costas AM, Lubner CE, Mulder DW, Tokmina-Lukaszewska M, Artz JH, Patterson A, Magnuson T, Jay ZJ, Duan HD, Miller J, Plunkett MH, Hoben JP, Barney BM, Carlson RP, Miller AF, Bothner B, King PW, Peters JW, Seefeldt LC (2017) The electron bifurcating FixABCX protein complex from Azotobactervinelandii: generation of low-potential reducing equivalents for nitrogenase catalysis. Biochemistry 56(32):4177–4190
Lenneman EM, Ohlert JM, Palani NP, Barney BM (2013) Fatty alcohols for wax esters in Marinobacter aquaeolei VT8: two optional routes in the wax biosynthesis pathway. Appl Environ Microbiol 79(22):7055–7062
Liew PWY, Jong BC, Najimudin N (2015) Hypothetical protein Avin_16040 as the S-layer protein of Azotobacter vinelandii and its involvement in plant root surface attachment. Appl Environ Microbiol 81(21):7484–7495
Mus F, Khokhani D, MacIntyre AM, Rugoli E, Dixon R, Ane JM, Peters JW (2022) Genetic determinants of ammonium excretion in nifL mutants of Azotobacter vinelandii. Appl Environ Microbiol 88(6):e0187621
Ortiz-Marquez JC, Do Nascimento M, Curatti L (2014) Metabolic engineering of ammonium release for nitrogen-fixing multispecies microbial cell-factories. Metab Eng 23:154–164
Ortiz-Marquez JC, Do Nascimento M, Dublan MD, Curatti L (2012) Association with an ammonium-excreting bacterium allows diazotrophic culture of oil-rich eukaryotic microalgae. Appl Environ Microbiol 78(7):2345–2352
Plunkett MH, Knutson CM, Barney BM (2020) Key factors affecting ammonium production by an Azotobacter vinelandii strain deregulated for biological nitrogen fixation. Microb Cell Fact 19(1):107
Roberts GP, Macneil T, Macneil D, Brill WJ (1978) Regulation and characterization of protein products coded by the nif (nitrogen fixation) genes of Klebsiella pneumonia. J Bacteriol 136(1):267–279
Ruvkun GB, Sundaresan V, Ausubel FM (1982) Directed transposon Tn5 mutagenesis and complementation analysis of Rhizobium meliloti symbiotic nitrogen fixation genes. Cell 29(2):551–559
Schipke CG, Goodin DB, McRee DE, Stout CD (1999) Oxidized and reduced Azotobacter vinelandii ferredoxin I at 1.4 Å resolution: conformational change of surface residues without significant change in the [3Fe-4S]+/0 cluster. Biochemistry 38(26):8228–8239
Schmehl M, Jahn A, Vilsendorf AMZ, Hennecke S, Masepohl B, Schuppler M, Marxer M, Oelze J, Klipp W (1993) Identification of a new class of nitrogen fixation genes in Rhodobacter capsulatus: a putative membrane complex involved in electron transport to nitrogenase. Mol Gen Genet 241(5–6):602–615
Segal HM, Spatzal T, Hill MG, Udit AK, Rees DC (2017) Electrochemical and structural characterization of Azotobacter vinelandii flavodoxin II. Protein Sci 26(10):1984–1993
Setubal JC, dos Santos P, Goldman BS, Ertesvåg H, Espin G, Rubio LM, Valla S, Almeida NF, Balasubramanian D, Cromes L, Curatti L, Du Z, Godsy E, Goodner B, Hellner-Burris K, Hernandez JA, Houmiel K, Imperial J, Kennedy C, Larson TJ, Latreille P, Ligon LS, Lu J, Maerk M, Miller NM, Norton S, O’Carroll IP, Paulsen I, Raulfs EC, Roemer R, Rosser J, Segura D, Slater S, Stricklin SL, Studholme DJ, Sun J, Viana CJ, Wallin E, Wang B, Wheeler C, Zhu H, Dean DR, Dixon R, Wood D (2009) Genome sequence of Azotobacter vinelandii, an obligate aerobe specialized to support diverse anaerobic metabolic processes. J Bacteriol 191(14):4534–4545
Smil V (2001) Enriching the earth : Fritz Haber, Carl Bosch, and the transformation of world food production. MIT Press, Cambridge, Mass
Temme K, Zhao DH, Voigt CA (2012) Refactoring the nitrogen fixation gene cluster from Klebsiella oxytoca. Proc Natl Acad Sci USA 109(18):7085–7090
Villa JA, Ray EE, Barney BM (2014) Azotobacter vinelandii siderophore can provide nitrogen to support the culture of the green algae Neochloris oleoabundans and Scenedesmus sp. BA032. FEMS Microbiol Lett 351:70–77
Funding
This work was supported by grants from the MnDRIVE transdisciplinary research initiative through the University of Minnesota based on funding from the state of Minnesota, the National Institute of Food and Agriculture (Project Numbers MIN-12–070 and MIN-12–081) and award number 2020–67019-31148 through the US Department of Agriculture. M. H. P. was further supported through funding from the National Science Foundation (CBET-1437758).
Author information
Authors and Affiliations
Contributions
B. M. B. conceived the study and designed experiments. B. M. B. and M. H. P. designed plasmids and constructed strains. M. H. P. and B. M. B. developed methods and collected data. All authors contributed to writing the manuscript and discussion.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barney, B.M., Plunkett, M.H. Rnf1 is the primary electron source to nitrogenase in a high-ammonium-accumulating strain of Azotobacter vinelandii. Appl Microbiol Biotechnol 106, 5051–5061 (2022). https://doi.org/10.1007/s00253-022-12059-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12059-x