Abstract
Purpose
To investigate the specific features of static functional connectivity (SFC) and dynamic functional connectivity (DFC) of networks related to cognition in patients with subcortical ischemic vascular disease (SIVD).
Methods
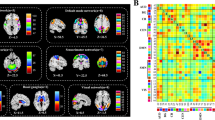
In this retrospective study, resting-state functional MRI data and a series of cognitive scores were obtained from 38 patients with SIVD and 23 normal controls. Independent component analysis, sliding window method, k-means clustering analysis and graph theory method were used to examine FC between the default mode network (DMN), dorsal attention network (DAN), frontoparietal network (FPN), salience network (SN) and executive control network (ECN) in patients with SIVD. Then, correlations between abnormal FC features and cognition were assessed.
Results
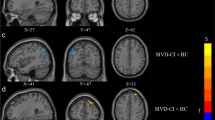
Compared with normal controls, SFC within the DMN significantly increased and SFC between the DMN and DAN significantly decreased in patients with SIVD. The decreased DFC mainly occurred in weakly connected states, especially the DFC of the SN; but the increased DFC, global network efficiency and local network efficiency and the decreased mean dwell time (MDT) and frequency mainly occurred in strongly connected states in SIVD patients. Moreover, aberrant SFC, DFC and MDT were significantly correlated with patients’ cognitive scores.
Conclusion
The overall results are suggestive of abnormal functional segregation and integration of SFC and DFC among networks related to cognition, especially in the SN. This may advance our comprehensive understanding of the abnormal changes in brain network connectivity in patients with SIVD. Our findings also highlight DFC may be an effective neuroimaging marker for the clinical diagnosis of SIVD.






Similar content being viewed by others
References
Román GC, Erkinjuntti T, Wallin A, Pantoni L, Chui HC (2002) Subcortical ischaemic vascular dementia. Lancet Neurol 1(7):426–436
Charlton RA, Morris RG, Nitkunan A, Markus HS (2006) The cognitive profiles of CADASIL and sporadic small vessel disease. Neurology 66(10):1523–1526
Sachdev P, Kalaria R, O’Brien J, Skoog I, Alladi S, Black SE et al (2014) Diagnostic criteria for vascular cognitive disorders: a VASCOG statement. Alzheimer Dis Assoc Disord 28(3):206–218
Wallin A, Roman GC, Esiri M, Kettunen P, Svensson J, Paraskevas GP et al (2018) Update on Vascular Cognitive Impairment Associated with Subcortical Small-Vessel Disease. J Alzheimers Dis 62(3):1417–1441
Ye Q, Bai F (2018) Contribution of diffusion, perfusion and functional MRI to the disconnection hypothesis in subcortical vascular cognitive impairment. Stroke Vasc Neurol 3(3):131–139
Wang R, Liu N, Tao YY, Gong XQ, Zheng J, Yang C et al (2020) The Application of rs-fMRI in Vascular Cognitive Impairment. Front Neurol 11:951
Damoiseaux JS, Rombouts SA, Barkhof F, Scheltens P, Stam CJ, Smith SM et al (2006) Consistent resting-state networks across healthy subjects. Proc Natl Acad Sci USA 103(37):13848–13853
Wei Y, de Lange S, Scholtens L, Watanabe K, Ardesch D, Jansen P et al (2019) Genetic map** and evolutionary analysis of human-expanded cognitive networks. Nat Commun 10(1):4839
Schulz M, Malherbe C, Cheng B, Thomalla G, Schlemm E (2021) Functional connectivity changes in cerebral small vessel disease - a systematic review of the resting-state MRI literature. BMC Med 19(1):103
Agosta F, Pievani M, Geroldi C, Copetti M, Frisoni GB, Filippi M (2012) Resting state fMRI in Alzheimer’s disease: beyond the default mode network. Neurobiol Aging 33(8):1564–1578
Dey AK, Stamenova V, Turner G, Black SE, Levine B (2016) Pathoconnectomics of cognitive impairment in small vessel disease: A systematic review. Alzheimers Dement 12(7):831–845
Ter Telgte A, van Leijsen EMC, Wiegertjes K, Klijn CJM, Tuladhar AM, de Leeuw FE (2018) Cerebral small vessel disease: from a focal to a global perspective. Nat Rev Neurol 14(7):387–398
Fu X, Liu F, Cui Z, Guo W (2020) Editorial dynamic functional connectivity in neuropsychiatric disorders: methods and applications. Front Neurosci 14:332
Christiaen E, Goossens MG, Descamps B, Larsen LE, Boon P, Raedt R et al (2020) Dynamic functional connectivity and graph theory metrics in a rat model of temporal lobe epilepsy reveal a preference for brain states with a lower functional connectivity, segregation and integration. Neurobiol Dis 139:104808
Wang Y, Wang C, Miao P, Liu J, Wei Y, Wu L et al (2020) An imbalance between functional segregation and integration in patients with pontine stroke: a dynamic functional network connectivity study. Neuroimage Clin 28:102507
Tu Y, Fu Z, Zeng F, Maleki N, Lan L, Li Z et al (2019) Abnormal thalamocortical network dynamics in migraine. Neurology 92(23):e2706–e2716
Espinoza FA, Liu J, Ciarochi J, Turner JA, Vergara VM, Caprihan A et al (2019) Dynamic functional network connectivity in Huntington’s disease and its associations with motor and cognitive measures. Hum Brain Mapp 40(6):1955–1968
Fu Z, Caprihan A, Chen J, Du Y, Adair JC, Sui J et al (2019) Altered static and dynamic functional network connectivity in Alzheimer’s disease and subcortical ischemic vascular disease: shared and specific brain connectivity abnormalities. Hum Brain Mapp 40(11):3203–3221
Li K, Fu Z, Luo X, Zeng Q, Huang P, Zhang MM et al (2020) The influence of cerebral small vessel disease on static and dynamic functional network connectivity in subjects along Alzheimer’s disease continuum. Brain Connect 11:189–200
Kim J, Criaud M, Cho SS, Diez-Cirarda M, Mihaescu A, Coakeley S et al (2017) Abnormal intrinsic brain functional network dynamics in Parkinson’s disease. Brain 140(11):2955–2967
Calhoun V, Adali T, Pearlson G, Pekar J (2001) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14(3):140–151
Rachakonda S, Silva R, Liu J, Calhoun V (2016) Memory efficient PCA methods for large group ICA. Front Neurosci 10:17
Himberg J, Hyvarinen A, Esposito F (2004) Validating the independent components of neuroimaging time series via clustering and visualization. Neuroimage 22(3):1214–1222
Bugli C, Lambert P (2007) Comparison between principal component analysis and independent component analysis in electroencephalograms modelling. Biomet J Biomet Zeitschrift 49(2):312–327
Allen EA, Erhardt EB, Damaraju E, Gruner W, Segall JM, Silva RF et al (2011) A baseline for the multivariate comparison of resting-state networks. Front Syst Neurosci 5:2
Damaraju E, Allen EA, Belger A, Ford JM, McEwen S, Mathalon DH et al (2014) Dynamic functional connectivity analysis reveals transient states of dysconnectivity in schizophrenia. Neuroimage Clin 5:298–308
Schumacher J, Peraza LR, Firbank M, Thomas AJ, Kaiser M, Gallagher P et al (2019) Dynamic functional connectivity changes in dementia with Lewy bodies and Alzheimer’s disease. Neuroimage Clin 22:101812
Zhang J, Wang J, Wu Q, Kuang W, Huang X, He Y et al (2011) Disrupted brain connectivity networks in drug-naive, first-episode major depressive disorder. Biol Psychiatry 70(4):334–342
Hashmi JA, Loggia ML, Khan S, Gao L, Kim J, Napadow V et al (2017) Dexmedetomidine disrupts the local and global efficiencies of large-scale brain networks. Anesthesiology 126(3):419–430
Liu R, Wu W, Ye Q, Gu Y, Zou J, Chen X et al (2019) Distinctive and pervasive alterations of functional brain networks in cerebral small vessel disease with and without cognitive impairment. Dement Geriatr Cogn Disord 47:55–67
Shaw EE, Schultz AP, Sperling RA, Hedden T (2015) Functional connectivity in multiple cortical networks is associated with performance across cognitive domains in older adults. Brain Connect 5(8):505–516
Spreng RN, Sepulcre J, Turner GR, Stevens WD, Schacter DL (2013) Intrinsic architecture underlying the relations among the default, dorsal attention, and frontoparietal control networks of the human brain. J Cogn Neurosci 25(1):74–86
Chen H, Li Y, Liu Q, Shi Q, Wang J, Shen H et al (2019) Abnormal interactions of the salience network, central executive network, and default-mode network in patients with different cognitive impairment loads caused by leukoaraiosis. Front Neur Circ 13:42
Chen Y, Wang J, Zhang J, Zhang T, Chen K, Fleisher A et al (2014) Aberrant functional networks connectivity and structural atrophy in silent lacunar infarcts: relationship with cognitive impairments. J Alzheimers Dis 42(3):841–850
Yi L, Wang J, Jia L, Zhao Z, Lu J, Li K et al (2012) Structural and functional changes in subcortical vascular mild cognitive impairment: a combined voxel-based morphometry and resting-state fMRI study. PLoS One 7(9):e44758
Liu X, Chen L, Cheng R, Luo T, Lv F, Fang W et al (2019) Altered functional connectivity in patients with subcortical ischemic vascular disease: A resting-state fMRI study. Brain Res 1715:126–133
Rashid B, Damaraju E, Pearlson GD, Calhoun VD (2014) Dynamic connectivity states estimated from resting fMRI Identify differences among Schizophrenia, bipolar disorder, and healthy control subjects. Front Hum Neurosci 8:897
Allen EA, Damaraju E, Plis SM, Erhardt EB, Eichele T, Calhoun VD (2014) Tracking whole-brain connectivity dynamics in the resting state. Cereb Cortex 24(3):663–676
Di X, Biswal BB (2014) Modulatory interactions between the default mode network and task positive networks in resting-state. PeerJ 2:e367
Goulden N, Khusnulina A, Davis NJ, Bracewell RM, Bokde AL, McNulty JP et al (2014) The salience network is responsible for switching between the default mode network and the central executive network: replication from DCM. Neuroimage 99:180–190
He X, Qin W, Liu Y, Zhang X, Duan Y, Song J et al (2014) Abnormal salience network in normal aging and in amnestic mild cognitive impairment and Alzheimer’s disease. Hum Brain Mapp 35(7):3446–3464
Park DC, Reuter-Lorenz P (2009) The adaptive brain: aging and neurocognitive scaffolding. Annu Rev Psychol 60:173–196
Barulli D, Stern Y (2013) Efficiency, capacity, compensation, maintenance, plasticity: emerging concepts in cognitive reserve. Trends Cogn Sci 17(10):502–509
Steffener J, Brickman AM, Rakitin BC, Gazes Y, Stern Y (2009) The impact of age-related changes on working memory functional activity. Brain Imaging Behav 3(2):142–153
Hutchison RM, Womelsdorf T, Allen EA, Bandettini PA, Calhoun VD, Corbetta M et al (2013) Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80:360–378
Acknowledgements
The authors thank the study participants.
Funding
This study was funded by the National Natural Science Foundation of China (Grant Nos. 81671666). National Natural Science Foundation of China, 81671666, Tianyou Luo.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, J., Cheng, R., Liu, X. et al. Abnormal static and dynamic functional connectivity of networks related to cognition in patients with subcortical ischemic vascular disease. Neuroradiology 64, 1201–1211 (2022). https://doi.org/10.1007/s00234-022-02895-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-022-02895-z