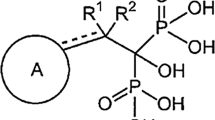
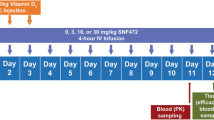
Abstract
Vascular calcification affects the prognosis of patients with renal failure. Bisphosphonates are regarded as candidate anti-calcifying drugs because of their inhibitory effects on both calcium-phosphate aggregation and bone resorption. However, calcification in well-known rodent models is dependent upon bone resorption accompanied by excessive bone turnover, making it difficult to estimate accurately the anti-calcifying potential of drugs. Therefore, models with low bone resorption are required to extrapolate anti-calcifying effects to humans. Three bisphosphonates (etidronate, alendronate, and FYB-931) were characterised for their inhibitory effects on bone resorption in vivo and calcium-phosphate aggregation estimated by calciprotein particle formation in vitro. Then, their effects were examined using two models inducing ectopic calcification: the site where lead acetate was subcutaneously injected into mice and the transplanted, aorta obtained from a donor rat. The inhibitory effects of bisphosphonates on bone resorption and calcium-phosphate aggregation were alendronate > FYB-931 > etidronate and FYB-931 > alendronate = etidronate, respectively. In the lead acetate-induced model, calcification was most potently suppressed by FYB-931, followed by alendronate and etidronate. In the aorta-transplanted model, only FYB-931 suppressed calcification at a high dose. In both the models, no correlation was observed between calcification and bone resorption marker, tartrate-resistant acid phosphatase (TRACP). Results from the lead acetate-induced model showed that inhibitory potency against calcium-phosphate aggregation contributed to calcification inhibition. The two calcification models, especially the lead acetate-induced model, may be ideal for the extrapolation of calcifying response to humans because of calcium-phosphate aggregation rather than bone resorption as its mechanism.






Similar content being viewed by others
References
Yamada S, Giachelli CM (2017) Vascular calcification in CKD-MBD: roles for phosphate, FGF23, and Klotho. Bone 100:87–93. https://doi.org/10.1016/j.bone.2016.11.012
Drake MT, Clarke BL, Khosla S (2008) Bisphosphonates: mechanism of action and role in clinical practice. Mayo Clin Proc 83:1032–1045. https://doi.org/10.4065/83.9.1032
Russell RG (2006) Bisphosphonates: from bench to bedside. Ann N Y Acad Sci 1068:367–401. https://doi.org/10.1196/annals.1346.041
Ariyoshi T, Eishi K, Sakamoto I, Matsukuma S, Odate T (2006) Effect of etidronic acid on arterial calcification in dialysis patients. Clin Drug Investig 26:215–222. https://doi.org/10.2165/00044011-200626040-00006
Pawade TA, Doris MK, Bing R, White AC, Forsyth L, Evans E, Graham C, Williams MC, van Beek EJR, Fletcher A, Adamson PD, Andrews JPM, Cartlidge TRG, Jenkins WSA, Syed M, Fujisawa T, Lucatelli C, Fraser W, Ralston SH, Boon N, Prendergast B, Newby DE, Dweck MR (2021) Effect of denosumab or alendronic acid on the progression of aortic stenosis: a double-blind randomized controlled trial. Circulation 143:2418–2427. https://doi.org/10.1161/CIRCULATIONAHA.121.053708
Yamada S, Tokumoto M, Tatsumoto N, Taniguchi M, Noguchi H, Nakano T, Masutani K, Ooboshi H, Tsuruya K, Kitazono T (2014) Phosphate overload directly induces systemic inflammation and malnutrition as well as vascular calcification in uremia. Am J Physiol Renal Physiol 306:F1418-1428. https://doi.org/10.1152/ajprenal.00633.2013
Tamagaki K, Yuan Q, Ohkawa H, Imazeki I, Moriguchi Y, Imai N, Sasaki S, Takeda K, Fukagawa M (2006) Severe hyperparathyroidism with bone abnormalities and metastatic calcification in rats with adenine-induced uraemia. Nephrol Dial Transplant 21:651–659. https://doi.org/10.1093/ndt/gfi273
Price PA, Faus SA, Williamson MK (2001) Bisphosphonates alendronate and ibandronate inhibit artery calcification at doses comparable to those that inhibit bone resorption. Arterioscler Thromb Vasc Biol 21:817–824. https://doi.org/10.1161/01.atv.21.5.817
Tamura K, Suzuki Y, Hashiba H, Tamura H, Aizawa S, Kogo H (2005) Effect of etidronate on aortic calcification and bone metabolism in calcitriol-treated rats with subtotal nephrectomy. J Pharmacol Sci 99:89–94. https://doi.org/10.1254/jphs.fpj05019x
Selye H, Gabbiani G, Serafimov N (1964) Histochemical studies on the role of the mast cell in calcergy. J Histochem Cytochem 12:563–569. https://doi.org/10.1177/12.8.563
Webb CL, Nguyen NM, Schoen FJ, Levy RJ (1992) Calcification of allograft aortic wall in a rat subdermal model. Pathophysiology and inhibition by Al3+ and aminodiphosphonate preincubations. Am J Pathol 141:487–496
Yamauchi H, Motomura N, Chung UI, Sata M, Takai D, Saito A, Ono M, Takamoto S (2013) Growth-associated hyperphosphatemia in young recipients accelerates aortic allograft calcification in a rat model. J Thorac Cardiovasc Surg 145:522–530. https://doi.org/10.1016/j.jtcvs.2012.03.010
Mathieu P, Roussel JC, Dagenais F, Anegon I (2003) Cartilaginous metaplasia and calcification in aortic allograft is associated with transforming growth factor beta 1 expression. J Thorac Cardiovasc Surg 126:1449–1454. https://doi.org/10.1016/s0022-5223(03)00031-x
Ishida K, Ashizawa N, Matsumoto K, Kobashi S, Kurita N, Shigematsu T, Iwanaga T (2019) Novel bisphosphonate compound FYB-931 preferentially inhibits aortic calcification in vitamin D3-treated rats. J Bone Miner Metab 37:796–804. https://doi.org/10.1007/s00774-019-00987-0
Ishida K, Ashizawa N, Morikane S, Kurita N, Kobashi S, Iwanaga T (2021) Assessment of calciprotein particle formation by AUC of the absorbance change: effect of FYB-931, a novel bisphosphonate compound. J Pharm Pharmacol 73:947–955. https://doi.org/10.1093/jpp/rgab019
Ferrer MD, Perez MM, Canaves MM, Buades JM, Salcedo C, Perello J (2017) A novel pharmacodynamic assay to evaluate the effects of crystallization inhibitors on calcium phosphate crystallization in human plasma. Sci Rep 7:6858. https://doi.org/10.1038/s41598-017-07203-x
Bridges JB, McClure J (1972) Experimental calcification in a number of species. Calcif Tissue Res 10:136–141. https://doi.org/10.1007/BF02012543
McClure J, Gardner DL (1976) The production of calcification in connective tissue and skeletal muscle using various chemical compounds. Calcif Tissue Res 22:129–135. https://doi.org/10.1007/BF02010352
Doyle DV, Dunn CJ, Willoughby DA (1979) Potassium permanganate induced calcergy: a model to study the effects of drugs on hydroxyapatite crystal deposition. J Pathol 128:63–69. https://doi.org/10.1002/path.1711280203
Kato Y, Takimoto S, Ogura H (1977) Mechanism of induction of hypercalcemia and hyperphosphatemia by lead acetate in the rat. Calcif Tissue Res 24:41–46. https://doi.org/10.1007/BF02223295
McClure J (1982) The effects of various anti-calcific, anti-rheumatic and anti-inflammatory drugs on local (simple) calcergy induced by lead acetate in the mouse. J Pathol 137:243–252. https://doi.org/10.1002/path.1711370308
Hirsch RS, McClure J, Vernon-Roberts B (1984) Induction and characterisation of local (simple) calcergy in granulation tissue in the rat by lead acetate injection into polyurethane sponges. Aust J Exp Biol Med Sci 62(Pt 6):727–742. https://doi.org/10.1038/icb.1984.69
Shiota E, Eguchi M, Yamaguchi T, Shimauchi T, Kuroki T, Shibata K, Kawano Y, Wada F, Kawamura H, Hashiguchi S (1982) The effects of ethane-1-hydroxy-1,1-diphosphonate (EHDP) on the local calcergy in rats. Orthop Traumatol 31:141–144. https://doi.org/10.5035/nishiseisai.31.141
Levy RJ, Hawley MA, Schoen FJ, Lund SA, Liu PY (1985) Inhibition by diphosphonate compounds of calcification of porcine bioprosthetic heart valve cusps implanted subcutaneously in rats. Circulation 71:349–356. https://doi.org/10.1161/01.cir.71.2.349
Levy RJ, Schoen FJ, Levy JT, Nelson AC, Howard SL, Oshry LJ (1983) Biologic determinants of dystrophic calcification and osteocalcin deposition in glutaraldehyde-preserved porcine aortic valve leaflets implanted subcutaneously in rats. Am J Pathol 113:143–155
Shankar R, Crowden S, Sallis JD (1984) Phosphocitrate and its analogue N-sulpho-2-amino tricarballylate inhibit aortic calcification. Atherosclerosis 52:191–198. https://doi.org/10.1016/0021-9150(84)90117-5
Amann K (2008) Media calcification and intima calcification are distinct entities in chronic kidney disease. Clin J Am Soc Nephrol 3:1599–1605. https://doi.org/10.2215/CJN.02120508
Tyson J, Bundy K, Roach C, Douglas H, Ventura V, Segars MF, Schwartz O, Simpson CL (2020) Mechanisms of the osteogenic switch of smooth muscle cells in vascular calcification: WNT signaling, BMPs, mechanotransduction, and EndMT. Bioengineering (Basel). https://doi.org/10.3390/bioengineering7030088
Lomashvili K, Garg P, O’Neill WC (2006) Chemical and hormonal determinants of vascular calcification in vitro. Kidney Int 69:1464–1470. https://doi.org/10.1038/sj.ki.5000297
Kawakami K, Ohya M, Yashiro M, Sonou T, Yamamoto S, Nakashima Y, Yano T, Tanaka Y, Ishida K, Kobashi S, Shigematsu T, Araki SI (2023) Bisphosphonate FYB-931 prevents high phosphate-induced vascular calcification in rat aortic rings by altering the dynamics of the transformation of calciprotein particles. Calcif Tissue Int 113:216–228. https://doi.org/10.1007/s00223-023-01086-z
Chen W, Anokhina V, Dieudonne G, Abramowitz MK, Kashyap R, Yan C, Wu TT, de Mesy Bentley KL, Miller BL, Bushinsky DA (2019) Patients with advanced chronic kidney disease and vascular calcification have a large hydrodynamic radius of secondary calciprotein particles. Nephrol Dial Transplant 34:992–1000. https://doi.org/10.1093/ndt/gfy117
Pasch A, Farese S, Graber S, Wald J, Richtering W, Floege J, Jahnen-Dechent W (2012) Nanoparticle-based test measures overall propensity for calcification in serum. J Am Soc Nephrol 23:1744–1752. https://doi.org/10.1681/ASN.2012030240
Bressendorff I, Hansen D, Schou M, Silver B, Pasch A, Bouchelouche P, Pedersen L, Rasmussen LM, Brandi L (2017) Oral magnesium supplementation in chronic kidney disease stages 3 and 4: efficacy, safety, and effect on serum calcification propensity-a prospective randomized double-blinded placebo-controlled clinical trial. Kidney Int Rep 2:380–389. https://doi.org/10.1016/j.ekir.2016.12.008
Bressendorff I, Hansen D, Schou M, Pasch A, Brandi L (2018) The effect of increasing dialysate magnesium on serum calcification propensity in subjects with end stage kidney disease: a randomized, controlled clinical trial. Clin J Am Soc Nephrol 13:1373–1380. https://doi.org/10.2215/CJN.13921217
Spiegel DM, Farmer B (2009) Long-term effects of magnesium carbonate on coronary artery calcification and bone mineral density in hemodialysis patients: a pilot study. Hemodial Int 13:453–459. https://doi.org/10.1111/j.1542-4758.2009.00364.x
Tzanakis IP, Stamataki EE, Papadaki AN, Giannakis N, Damianakis NE, Oreopoulos DG (2014) Magnesium retards the progress of the arterial calcifications in hemodialysis patients: a pilot study. Int Urol Nephrol 46:2199–2205. https://doi.org/10.1007/s11255-014-0751-9
Sakaguchi Y, Hamano T, Obi Y, Monden C, Oka T, Yamaguchi S, Matsui I, Hashimoto N, Matsumoto A, Shimada K, Takabatake Y, Takahashi A, Kaimori JY, Moriyama T, Yamamoto R, Horio M, Yamamoto K, Sugimoto K, Rakugi H, Isaka Y (2019) A randomized trial of magnesium oxide and oral carbon adsorbent for coronary artery calcification in predialysis CKD. J Am Soc Nephrol 30:1073–1085. https://doi.org/10.1681/ASN.2018111150
Acknowledgements
We thank Dr. Koji Matsumoto, and Ms. Noriko Chikamatsu in FUJI YAKUHIN CO., LTD. for their assistance in the in vivo evaluation.
Funding
The authors are employees of FUJI YAKUHIN CO., LTD.
Author information
Authors and Affiliations
Contributions
Participated in research design: Shota Morikane, Koichi Ishida, Naoki Ashizawa, Naoki Kurita, Seiichi Kobashi and Takashi Iwanaga. Conducted experiments: Shota Morikane, Koichi Ishida and Naoki Ashizawa. Wrote or contributed to the writing of the manuscript: Shota Morikane, Naoki Ashizawa, Koichi Ishida, Masaya Matsubayashi, and Tetsuya Taniguchi. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Shota Morikane, Koichi Ishida, Naoki Ashizawa, Tetsuya Taniguchi, Masaya Matsubayashi, Naoki Kurita, Seiichi Kobashi and, Takashi Iwanaga have no conflict of interest directly relevant to the content of this manuscript.
Human and animal rights
The animal experiments were approved by the Animal Care Utilisation Committee of FUJI YAKUHIN Research Laboratories, which is accredited by the Japan Health Sciences Foundation. All procedures were performed in compliance with ARRIVE and the National Guidelines for Animal Care and Use.
Informed consent statements
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morikane, S., Ishida, K., Ashizawa, N. et al. Lead Acetate-Injected Mice is an Animal Model for Extrapolation of Calcifying Response to Humans Due to Low Involvement of Bone Resorption. Calcif Tissue Int (2024). https://doi.org/10.1007/s00223-024-01245-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00223-024-01245-w