Abstract
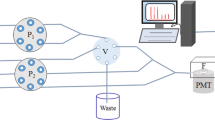
A novel chemiluminescence (CL) system based on the reaction of fluorescent water-soluble nitrogen-rich quantum dots (N-dots) and diperiodatoargentate(III) (DPA) was developed. The prepared N-dots have a small size (≤10 nm) and high percentage of nitrogen (39.9 %), which exceeds the content of carbon in the same N-dots. The N-dots exhibit characteristic blue fluorescence under UV light and up-conversion luminescence. The relatively intense CL emission is based on the direct oxidation of N-dots by DPA. The CL emission may be attributed to the high nitrogen content and the special structure of the N-dots. The CL mechanism of N-dots and DPA was investigated by using CL, UV–Vis absorption, IR, fluorescence, and radical scavenging experiments. This investigation provides a way to study the optical properties of N-dots. The analytical applicability of the N-dots and DPA CL system in the determination of ferulic acid (FA) was explored. The CL intensity was linearly proportional to the concentration of ferulic acid from 3.0 × 10−7 to 1.0 × 10−5 g mL−1 with a detection limit of 8.0 × 10−8 g mL−1 (3σ); the relative standard deviation was 2.4 % for 4.0 × 10−7 g mL−1 FA (n = 9). The proposed method was successfully applied to the determination of ferulic acid in Angelica sinensis. The study provides valuable insight into the role of nitrogen-rich quantum dots in CL.








Similar content being viewed by others
References
Lin Z, Chen H, Lin J. Peroxide induced ultra-weak chemiluminescence and its application in analytical chemistry. Analyst. 2013;138(18):5182–93.
Iranifam M. Revisiting flow-chemiluminescence techniques: pharmaceutical analysis. Luminescence. 2013;28(6):798–820.
Christodouleas D, Fotakis C, Economou A, Papadopoulos K, Timotheou-Potamia M, Calokerinos A. Flow-based methods with chemiluminescence detection for food and environmental analysis: a review. Anal Lett. 2011;44(1-3):176–215.
Ocaña-González JA, Ramos-Payán M, Fernández-Torres R, Villar Navarro M, Bello-López MÁ. Application of chemiluminescence in the analysis of wastewaters – a review. Talanta. 2014;122:214–22.
Su Y, **e Y, Hou X, Lv Y. Recent advances in analytical applications of nanomaterials in liquid-phase chemiluminescence. Appl Spectrosc Rev. 2014;49(3):201–32.
Sanderson K. Quantum dots go large. Nature. 2009;459(7248):760–1.
Gao X, Cui Y, Levenson RM, Chung LWK, Nie S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol. 2004;22(8):969–76.
Chen H, Lin L, Li H, Lin J. Quantum dots-enhanced chemiluminescence: mechanism and application. Coord Chem Rev. 2014;263:86–100.
Huang X, Li L, Qian H, Dong C, Ren J. A resonance energy transfer between chemiluminescent donors and luminescent quantum-dots as acceptors (CRET). Angew Chem Int Ed. 2006;45:5140–3.
Dong S, Liu F, Lu C. Organo-modified hydrotalcite-quantum dot nanocomposites as a novel chemiluminescence resonance energy transfer probe. Anal Chem. 2013;85(6):3363–8.
Zhou W, Cao Y, Sui D, Lu C. Radical pair-driven luminescence of quantum dots for specific detection of peroxynitrite in living cells. Anal Chem. 2016;88(5):2659–65.
Derfus AM, Chan WCW, Bhatia SN. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004;4(1):11–8.
Bhunia SK, Pradhan N, Jana NR. Vitamin B1 derived blue and green fluorescent carbon nanoparticles for cell-imaging application. ACS Appl Mater Interfaces. 2014;6(10):7672–9.
Zheng X, Ananthanarayanan A, Luo K, Chen P. Glowing graphene quantum dots and carbon dots: properties, syntheses, and biological applications. Small. 2015;11(14):1620–36.
Li H, Kang Z, Liu Y, Lee S. Carbon nanodots: synthesis, properties and applications. J Mater Chem. 2012;22(46):24230–53.
Yao Q, Lin L, Zhao T, Chen X. Advances in preparation, physicochemical properties and applications of heteroatom-doped graphene quantum dots. Prog Chem. 2015;27(11):1523–30.
Zhang Y, Ma D, Zhuang Y, Zhang X, Chen W, Hong L, et al. One-pot synthesis of N-doped carbon dots with tunable luminescence properties. J Mater Chem. 2012;22(33):16714–8.
Zhu S, Meng Q, Wang L, Zhang J, Song Y, ** H, et al. Highly photoluminescent carbon dots for multicolor patterning, sensors, and bioimaging. Angew Chem Int Ed. 2013;52(14):3953–7.
Hu S, Tian R, Dong Y, Yang J, Liu J, Chang Q. Modulation and effects of surface groups on photoluminescence and photocatalytic activity of carbon dots. Nanoscale. 2013;5(23):11665–71.
Ke Y, Garg B, Ling Y. Waste chicken eggshell as low-cost precursor for efficient synthesis of nitrogen-doped fluorescent carbon nanodots and their multi-functional applications. RSC Adv. 2014;4(102):58329–36.
Xu H, Zhou S, **ao L, Wang H, Li S, Yuan Q. Fabrication of a nitrogen-doped graphene quantum dot from MOF-derived porous carbon and its application for highly selective fluorescence detection of Fe3+. J Mater Chem C. 2015;3(2):291–7.
Liang Q, Ma W, Shi Y, Li Z, Yang X. Easy synthesis of highly fluorescent carbon quantum dots from gelatin and their luminescent properties and applications. Carbon. 2013;60:421–8.
Xu Y, Wu M, Liu Y, Feng X, Yin X, He X, et al. Nitrogen-doped carbon dots: a facile and general preparation method, photoluminescence investigation, and imaging applications. Chem Eur J. 2013;19(7):2276–83.
Hu C, Liu Y, Yang Y, Cui J, Huang Z, Wang Y, et al. One-step preparation of nitrogen-doped graphene quantum dots from oxidized debris of graphene oxide. J Mater Chem B. 2013;1(1):39–42.
Dong Y, Pang H, Yang HB, Guo C, Shao J, Chi Y, et al. Carbon-based dots Co-doped with nitrogen and sulfur for high quantum yield and excitation-independent emission. Angew Chem Int Ed. 2013;52(30):7800–4.
Amjadi M, Manzoori JL, Hallaj T. Chemiluminescence of graphene quantum dots and its application to the determination of uric acid. J Lumin. 2014;153:73–8.
Hallaj T, Amjadi M, Manzoori JL, Shokri R. Chemiluminescence reaction of glucose-derived graphene quantum dots with hypochlorite, and its application to the determination of free chlorine. Microchim Acta. 2015;182(3-4):789–96.
Teng P, **e J, Long Y, Huang X, Zhu R, Wang X, et al. Chemiluminescence behavior of the carbon dots and the reduced state carbon dots. J Lumin. 2014;146:464–9.
Amjadi M, Manzoori JL, Hallaj T, Sorouraddin MH. Direct chemiluminescence of carbon dots induced by potassium ferricyanide and its analytical application. Spectrochim Acta A Mol Biomol Spectrosc. 2014;122:715–20.
Shi H, Guo Y, Kang W. Kinetic study of the oxidation of N, N-dimethylethanolamine by bis(hydrogenperiodato)argentate(III) in aqueous solution. J Solution Chem. 2011;40(7):1371–81.
Fu Z, Li G, Hu Y. A novel flow injection chemiluminescence method for the determination of indole-3-acetic acid in biological samples by using trivalent silver. Anal Methods. 2015;7(11):4590–5.
Sun H, Chen P, Wang F, Wen H. Investigation on enhanced chemiluminescence reaction systems with bis(hydrogenperiodato)argentate(III) complex anion for fluoroquinolones synthetic antibiotics. Talanta. 2009;79(2):134–40.
Chen X, ** Q, Wu L, Tung C, Tang X. Synthesis and unique photoluminescence properties of nitrogen-rich quantum dots and their applications. Angew Chem Int Ed. 2014;46(53):12542–7.
Balikungeri A, Pelletier M, Monnier D. Contribution to the study of the complexes bis(dihydrogen tellurato)cuprate(III) and argentate(III), bis(hydrogen periodato)cuprate(III) and argentate(III). Inorg Chim Acta. 1977;22:7–14.
Da Silva JCGE, Gonçalves HMR. Analytical and bioanalytical applications of carbon dots. TrAC Trends Anal Chem. 2011;30(8):1327–36.
Chen W, Joly AG, McCready DE. Upconversion luminescence from CdSe nanoparticles. J Chem Phys. 2005;122(22):224708.
Zhao L, Di F, Wang D, Guo L, Yang Y, Wan B, et al. Chemiluminescence of carbon dots under strong alkaline solutions: a novel insight into carbon dot optical properties. Nanoscale. 2013;5(7):2655–8.
Shi H, Shen S, Sun H, Liu Z, Li L. Oxidation of l-serine and l-threonine by bis(hydrogen periodato)argentate(III) complex anion: a mechanistic study. J Inorg Biochem. 2007;101(1):165–72.
Peng C, Hsieh C, Wang H, Chung J, Chen K, Peng RY. Ferulic acid is nephrodamaging while gallic acid is renal protective in long term treatment of chronic kidney disease. Clin Nutr. 2012;31(3):405–14.
Luo L, Wang X, Li Q, Ding Y, Jia J, Deng D. Voltammetric determination of ferulic acid didodecyldimethylammonium bromide/nafion composite film-modified carbon paste electrode. Anal Sci. 2010;26(8):907–11.
Acknowledgments
The work were supported by the National Natural Science Foundation of China (nos. 21475153 and 21575167), the Guangdong Provincial Natural Science Foundation of China (No. 2015A030311020), and the Special funds for public welfare research and capacity building in Guangdong Province of China (no. 2015A030401036), respectively.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Published in the topical collection Highlights of Analytical Chemical Luminescence with guest editors Aldo Roda, Hua Cui, and Chao Lu.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 264 kb)
Rights and permissions
About this article
Cite this article
Fu, Z., Li, G. & Hu, Y. Chemiluminescence of nitrogen-rich quantum dots in diperiodatoargentate(III) solution and its application in ferulic acid analysis. Anal Bioanal Chem 408, 8813–8820 (2016). https://doi.org/10.1007/s00216-016-9786-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9786-3