Abstract
Rationale
Chronic stress exposure disrupts the medial prefrontal cortex’s (mPFC) ability to regulate impulses, leading to the loss of control over alcohol drinking in rodents, emphasizing the critical role of this forebrain area in regulating alcohol consumption. Moreover, chronic stress exposure causes lateralization of mPFC functions with volumetric and functional changes, resulting in hyperactivity in the right hemisphere and functional decrease in the left.
Objectives
This study investigated the inhibitory role of the left prelimbic cortex (LPrL) on ethanol consumption induced by chronic social defeat stress (SDS) in male mice and to examine if inactivation of the LPrL causes disinhibition of the right mPFC, leading to an increase in ethanol consumption. We also investigated the role of lateralization and neurochemical alterations in the mPFC related to ethanol consumption induced by chronic SDS. To this end, we examined the activation patterns of ΔFosB, VGLUT2, and GAD67 in the left and right mPFC.
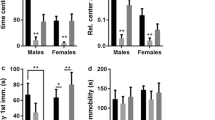
Results
Temporarily blocking the LPrL or right PrL (RPrL) cortices during acute SDS did not affect male mice’s voluntary ethanol consumption in male mice. When each cortex was blocked in mice previously exposed to chronic SDS, ethanol consumption also remained unaffected. However, male mice with LPrL lesions during chronic SDS showed an increase in voluntary ethanol consumption, which was associated with enhanced ΔFosB/VGLUT2-positive neurons within the RPrL cortex.
Conclusions
The results suggest that the LPrL may play a role in inhibiting ethanol consumption induced by chronic SDS, while the RPrL may be involved in the disinhibition of ethanol consumption.






Similar content being viewed by others
References
Anacker AMJ, Ryabinin AE (2010) Biological contribution to social influences on alcohol drinking: evidence from animal models Int J Environ Res Public Health 7:473–493 https://doi.org/10.3390/ijerph7020473
Bahi A (2017) Environmental enrichment reduces chronic psychosocial stress-induced anxiety and ethanol-related behaviors in mice Prog Neuro-Psychopharmacology Biol Psychiatry 77:65–74 https://doi.org/10.1016/j.pnpbp.2017.04.001
Bahi A, Dreyer J-L (2023) Anxiety and ethanol consumption in socially defeated mice; effect of hippocampal serotonin transporter knockdown Behav Brain Res 451:114508 https://doi.org/10.1016/j.bbr.2023.114508
Bechara A, Damasio H (2002) Decision-making and addiction (part I): impaired activation of somatic states in substance dependent individuals when pondering decisions with negative future consequences Neuropsychologia 40:1675–1689 https://doi.org/10.1016/S0028-3932(02)00015-5
Becker HC, Lopez MF, Doremus-Fitzwater TL (2011) Effects of stress on alcohol drinking: a review of animal studies Psychopharmacology 218:131–156 https://doi.org/10.1007/s00213-011-2443-9
Belknap JK, Crabbe JC, Young ER (1993) Voluntary consumption of ethanol in 15 inbred mouse strains Psychopharmacology 112:503–510 https://doi.org/10.1007/BF02244901
Breese GR, Sinha R, Heilig M (2011) Chronic alcohol neuroadaptation and stress contribute to susceptibility for alcohol craving and relapse Pharmacol Ther 129(2):149–71
Burgess A, Vigneron S, Brioudes E et al (2010) Loss of human Greatwall results in G2 arrest and multiple mitotic defects due to deregulation of the cyclin B-Cdc2/PP2A balance Proc Natl Acad Sci 107:12564–12569 https://doi.org/10.1073/pnas.0914191107
Canto-de-Souza L, Mattioli R (2016) The consolidation of inhibitory avoidance memory in mice depends on the intensity of the aversive stimulus: the involvement of the amygdala, dorsal hippocampus and medial prefrontal cortex Neurobiol Learn Mem 130:44–51 https://doi.org/10.1016/j.nlm.2016.01.012
Canto-de-Souza L, Demetrovich PG, Plas S et al (2021) Daily optogenetic stimulation of the left infralimbic cortex reverses extinction impairments in male rats exposed to single prolonged stress Front Behav Neurosci 15:780326 https://doi.org/10.3389/fnbeh.2021.780326
Carlson JN, Drew Stevens K (2006) Individual differences in ethanol self-administration following withdrawal are associated with asymmetric changes in dopamine and serotonin in the medial prefrontal cortex and amygdala Alcohol Clin Exp Res https://doi.org/10.1111/j.1530-0277.2006.00203.x
Caruso MJ, Seemiller LR, Fetherston TB et al (2018) Adolescent social stress increases anxiety-like behavior and ethanol consumption in adult male and female C57BL/6J mice Sci Rep 8:10040 https://doi.org/10.1038/s41598-018-28381-2
Cerqueira JJ, Almeida OFX, Sousa N (2008) The stressed prefrontal cortex. Left? Right! Brain Behav Immun 22:630–638
Costa NS, Vicente MA, Cipriano AC et al (2016) Functional lateralization of the medial prefrontal cortex in the modulation of anxiety in mice: left or right? Neuropharmacology 108:82–90 https://doi.org/10.1016/j.neuropharm.2016.04.011
Crestani CC, Alves FHF, Tavares RF, Corrêa FMA (2009) Role of the bed nucleus of the stria terminalis in the cardiovascular responses to acute restraint stress in rats. Stress 12:268–278 https://doi.org/10.1080/10253890802331477
Croft AP, Brooks SP, Cole J, Little HJ (2005) Social defeat increases alcohol preference of C57BL/10 strain mice; effect prevented by a CCKB antagonist. Psychopharmacology 183:163–170 https://doi.org/10.1007/s00213-005-0165-6
Cruz FC, Quadros IM, da S. Planeta C, KA, Miczek (2008) Maternal separation stress in male mice: long-term increases in alcohol intake Psychopharmacology 201:459–468 https://doi.org/10.1007/s00213-008-1307-4
Czéh B, Müller-Keuker JIH, Rygula R et al (2007) Chronic social stress inhibits cell proliferation in the adult medial prefrontal cortex: hemispheric asymmetry and reversal by fluoxetine treatment. Neuropsychopharmacology 32:1490–1503 https://doi.org/10.1038/sj.npp.1301275
Damasio AR (2000) The fabric of the mind: a neurobiological perspective In: Progress in Brain Research pp 457–467
Dent G, Choi DC, Herman JP, Levine S (2007) GABAergic circuits and the stress hyporesponsive period in the rat: ontogeny of glutamic acid decarboxylase (GAD) 67 mRNA expression in limbic–hypothalamic stress pathways Brain Res 1138:1–9 https://doi.org/10.1016/j.brainres.2006.04.082
Euston DR, Gruber AJ, McNaughton BL (2012) The role of medial prefrontal cortex in memory and decision making Neuron 76:1057–1070 https://doi.org/10.1016/j.neuron.2012.12.002
Fadda F, Rossetti ZL (1998) Chronic ethanol consumption: from neuroadaptation to neurodegeneration Prog Neurobiol 56:385–431 https://doi.org/10.1016/s0301-0082(98)00032-x
Faria MP, Laverde CF, Nunes-de-Souza RL (2020) Anxiogenesis induced by social defeat in male mice: role of nitric oxide, NMDA, and CRF1 receptors in the medial prefrontal cortex and BNST Neuropharmacology 166:107973 https://doi.org/10.1016/j.neuropharm.2020.107973
Franklin KBJ Paxinos G 2007 The mouse brain in stereotaxic coordinates (map)
Fremeau RT, Troyer MD, Pahner I et al (2001) The expression of vesicular glutamate transporters defines two classes of excitatory synapse Neuron 31:247–260 https://doi.org/10.1016/S0896-6273(01)00344-0
Ghosal S, Duman CH, Liu R-J et al (2020) Ketamine rapidly reverses stress-induced impairments in GABAergic transmission in the prefrontal cortex in male rodents Neurobiol Dis 134:104669 https://doi.org/10.1016/j.nbd.2019.104669
Gilabert-Juan J, Castillo-Gomez E, Pérez-Rando M et al (2011) Chronic stress induces changes in the structure of interneurons and in the expression of molecules related to neuronal structural plasticity and inhibitory neurotransmission in the amygdala of adult mice. Exp Neurol 232:33–40 https://doi.org/10.1016/j.expneurol.2011.07.009
Gilabert-Juan J, Castillo-Gomez E, Guirado R et al (2013) Chronic stress alters inhibitory networks in the medial prefrontal cortex of adult mice Brain Struct Funct https://doi.org/10.1007/s00429-012-0479-1
Gill K, Liu Y, Deitrich RA (1996) Voluntary alcohol consumption in BXD recombinant inbred mice: relationship to alcohol metabolism Alcohol Clin Exp Res 20:185–190 https://doi.org/10.1111/j.1530-0277.1996.tb01063.x
Glick SD, Merski C, Steindorf S et al (1992) Neurochemical predisposition to self-administer morphine in rats Brain Res 578:215–220 https://doi.org/10.1016/0006-8993(92)90250-D
Golden SA, Covington HE, Berton O, Russo SJ (2011) A standardized protocol for repeated social defeat stress in mice Nat Protoc 6:1183–1191 https://doi.org/10.1038/nprot.2011.361
Goldman D, Oroszi G, Ducci F (2005) The genetics of addictions: uncovering the genes Nat Rev Genet 6:521–532 https://doi.org/10.1038/nrg1635
Hadad-Ophir O, Albrecht A, Stork O, Richter-Levin G (2014) Amygdala activation and GABAergic gene expression in hippocampal sub-regions at the interplay of stress and spatial learning Front Behav Neurosci 8 https://doi.org/10.3389/fnbeh.2014.00003
Haun HL, Griffin WC, Lopez MF et al (2018) Increasing brain-derived neurotrophic factor (BDNF) in medial prefrontal cortex selectively reduces excessive drinking in ethanol dependent mice Neuropharmacology 140:35–42 https://doi.org/10.1016/j.neuropharm.2018.07.031
Heilig M Koob GF (2007) A key role for corticotropin-releasing factor in alcohol dependence Trends Neurosci
Holmes A, Fitzgerald PJ, MacPherson KP et al (2012) Chronic alcohol remodels prefrontal neurons and disrupts NMDAR-mediated fear extinction encoding Nat Neurosci 15:1359–1361 https://doi.org/10.1038/nn.3204
Johnstone T, van Reekum CM, Urry HL et al (2007) Failure to regulate: counterproductive recruitment of top-down prefrontal-subcortical circuitry in major depression J Neurosci 27:8877–8884 https://doi.org/10.1523/JNEUROSCI.2063-07.2007
Kesner RP, Churchwell JC (2011) An analysis of rat prefrontal cortex in mediating executive function Neurobiol Learn Mem 96:417–431 https://doi.org/10.1016/j.nlm.2011.07.002
Krettek JE, Price JL (1977) The cortical projections of the mediodorsal nucleus and adjacent thalamic nuclei in the rat J Comp Neurol 171:157–191 https://doi.org/10.1002/cne.901710204
Kretz R (1984) Local cobalt injection: a method to discriminate presynaptic axonal from postsynaptic neuronal activity J Neurosci Methods 11:129–135 https://doi.org/10.1016/0165-0270(84)90030-X
Kroener S, Mulholland PJ, New NN et al (2012) Chronic alcohol exposure alters behavioral and synaptic plasticity of the rodent prefrontal cortex PLoS ONE 7:e37541 https://doi.org/10.1371/journal.pone.0037541
LaLumiere RT (2014) Optogenetic dissection of amygdala functioning Front Behav Neurosci 8 https://doi.org/10.3389/fnbeh.2014.00107
Lee E, Hong J, Park Y-G et al (2015) Left brain cortical activity modulates stress effects on social behavior Sci Rep 5:13342 https://doi.org/10.1038/srep13342
Lei K, Wegner SA, Yu JH et al (2016) Nucleus accumbens shell and mPFC but not insula orexin-1 receptors promote excessive alcohol drinking Front Neurosci 10 https://doi.org/10.3389/fnins.2016.00400
Liu R, Jolas T, Aghajanian G (2000) Serotonin 5-HT2 receptors activate local GABA inhibitory inputs to serotonergic neurons of the dorsal raphe nucleus Brain Res 873:34–45 https://doi.org/10.1016/S0006-8993(00)02468-9
Lomber SG (1999) The advantages and limitations of permanent or reversible deactivation techniques in the assessment of neural function J Neurosci Methods 86:109–117 https://doi.org/10.1016/S0165-0270(98)00160-5
Lu Y-L, Richardson HN (2014) Alcohol, stress hormones, and the prefrontal cortex: a proposed pathway to the dark side of addiction Neuroscience 277:139–151 https://doi.org/10.1016/j.neuroscience.2014.06.053
Macedo GC, Morita GM, Domingues LP et al (2018) Consequences of continuous social defeat stress on anxiety- and depressive-like behaviors and ethanol reward in mice Horm Behav 97:154–161 https://doi.org/10.1016/j.yhbeh.2017.10.007
Makinson R, Lundgren KH, Seroogy KB, Herman JP (2015) Chronic social subordination stress modulates glutamic acid decarboxylase (GAD) 67 mRNA expression in central stress circuits Physiol Behav 146:7–15 https://doi.org/10.1016/j.physbeh.2015.04.025
McBride WJ, Rodd ZA, Bell RL et al (2014) The alcohol-preferring (P) and high-alcohol-drinking (HAD) rats – animal models of alcoholism Alcohol 48:209–215 https://doi.org/10.1016/j.alcohol.2013.09.044
McKlveen JM, Myers B, Herman JP (2015) The medial prefrontal cortex: coordinator of autonomic, neuroendocrine and behavioural responses to stress J Neuroendocrinol 27:446–456 https://doi.org/10.1111/jne.12272
McKlveen JM, Moloney RD, Scheimann JR et al (2019) “Braking” the prefrontal cortex: the role of glucocorticoids and interneurons in stress adaptation and pathology Biol Psychiatry 86:669–681 https://doi.org/10.1016/j.biopsych.2019.04.032
Mendes-Gomes J, Amaral VCS, Nunes-de-Souza RL (2011) Ventrolateral periaqueductal gray lesion attenuates nociception but does not change anxiety-like indices or fear-induced antinociception in mice Behav Brain Res 219:248–253 https://doi.org/10.1016/j.bbr.2011.01.012
Miczek KA, Thompson ML, Shuster L (1982) Opioid-like analgesia in defeated mice Science 215:1520–1522 https://doi.org/10.1126/science.7199758
Moghaddam B, Homayoun H (2008) Divergent plasticity of prefrontal cortex networks Neuropsychopharmacology 33:42–55 https://doi.org/10.1038/sj.npp.1301554
Moselhy HF, Georgiou G, Kahn A (2001) Frontal lobe changes in alcoholism: a review of the literature Alcohol Alcohol 36:357–368 https://doi.org/10.1093/alcalc/36.5.357
Nestler EJ (2008) Transcriptional mechanisms of addiction: role of ΔFosB Philos Trans R Soc B Biol Sci 363 https://doi.org/10.1098/rstb.2008.0067
Nielsen DM, Crosley KJ, Keller RW et al (1999) Left and right 6-hydroxydopamine lesions of the medial prefrontal cortex differentially affect voluntary ethanol consumption Brain Res 823:59–66 https://doi.org/10.1016/S0006-8993(99)01099-9
Norman KJ, Seiden JA, Klickstein JA et al (2015) Social stress and escalated drug self-administration in mice I. Alcohol and Corticosterone Psychopharmacology (berl) 232:991–1001 https://doi.org/10.1007/s00213-014-3733-9
Ohta K, Suzuki S, Warita K et al (2020) The effects of early life stress on the excitatory/inhibitory balance of the medial prefrontal cortex Behav Brain Res 379:112306 https://doi.org/10.1016/j.bbr.2019.112306
Oppong-Damoah A, Blough BE, Makriyannis A, Murnane KS (2019) The sesquiterpene beta-caryophyllene oxide attenuates ethanol drinking and place conditioning in mice. Heliyon 5:e01915. https://doi.org/10.1016/j.heliyon.2019.e01915
Peluffo H, Acarin L, Arís A et al (2006) Neuroprotection from NMDA excitotoxic lesion by Cu/Zn superoxide dismutase gene delivery to the postnatal rat brain by a modular protein vector BMC Neurosci 7:35 https://doi.org/10.1186/1471-2202-7-35
Pleil KE, Lowery-Gionta EG, Crowley NA et al (2015) Effects of chronic ethanol exposure on neuronal function in the prefrontal cortex and extended amygdala Neuropharmacology 99:735–749 https://doi.org/10.1016/j.neuropharm.2015.06.017
Radke AK, Pickens CL, Holmes A (2014) The effects of stress on measures of alcohol drinking in rodents In: Neurobiology of alcohol dependence Elsevier pp 97–110
Rodriguez-Arias M, Navarrete F, Blanco-Gandia MC et al (2016) Social defeat in adolescent mice increases vulnerability to alcohol consumption Addict Biol 21:87–97 https://doi.org/10.1111/adb.12184
Rosenthal A, Beck A, Zois E et al (2019) Volumetric prefrontal cortex alterations in patients with alcohol dependence and the involvement of self-control Alcohol Clin Exp Res 43:2514–2524 https://doi.org/10.1111/acer.14211
Santos-Costa N, Baptista-de-Souza D, Canto-de-Souza L et al (2021) Glutamatergic neurotransmission controls the functional lateralization of the mPFC in the modulation of anxiety induced by social defeat stress in male mice. Front Behav Neurosci 15 https://doi.org/10.3389/fnbeh.2021.695735
Scopinho AA, Scopinho M, Lisboa SF et al (2010) Acute reversible inactivation of the ventral medial prefrontal cortex induces antidepressant-like effects in rats Behav Brain Res 214:437–442 https://doi.org/10.1016/j.bbr.2010.06.018
Sinha R, Fox HC, Hong KA et al (2009) Enhanced negative emotion and alcohol craving, and altered physiological responses following stress and cue exposure in alcohol dependent individuals Neuropsychopharmacology 34:1198–1208 https://doi.org/10.1038/npp.2008.78
Song I, Volynski K, Brenner T et al (2013) Different transporter systems regulate extracellular GABA from vesicular and non-vesicular sources Front Cell Neurosci 7 https://doi.org/10.3389/fncel.2013.00023
Steketee JD (2003) Neurotransmitter systems of the medial prefrontal cortex: potential role in sensitization to psychostimulants Brain Res Rev
Stephens DN, Duka T (2008) Cognitive and emotional consequences of binge drinking: role of amygdala and prefrontal cortex Philos Trans R Soc B Biol Sci 363:3169–3179 https://doi.org/10.1098/rstb.2008.0097
Sullivan RM, Gratton A (1999) Lateralized effects of medial prefrontal cortex lesions on neuroendocrine and autonomic stress responses in rats J Neurosci 19:2834–2840 https://doi.org/10.1523/JNEUROSCI.19-07-02834.1999
Tavares RFF, Corrêa FMAMA (2006) Role of the medial prefrontal cortex in cardiovascular responses to acute restraint in rats Neuroscience 143:231–240 https://doi.org/10.1016/j.neuroscience.2006.07.030
Uylings HBM, Groenewegen HJ, Kolb B (2003) Do rats have a prefrontal cortex? Behav Brain Res 146:3–17 https://doi.org/10.1016/j.bbr.2003.09.028
Vialou V, Bagot RC, Cahill ME et al (2014) Prefrontal cortical circuit for depression- and anxiety-related behaviors mediated by cholecystokinin: role of ΔFosB J Neurosci 34:3878–3887 https://doi.org/10.1523/JNEUROSCI.1787-13.2014
Victoriano G, Santos-Costa N, Mascarenhas DC, Nunes-de-Souza RL (2020) Inhibition of the left medial prefrontal cortex (mPFC) prolongs the social defeat-induced anxiogenesis in mice: attenuation by NMDA receptor blockade in the right mPFC Behav Brain Res 378:112312 https://doi.org/10.1016/j.bbr.2019.112312
WHO (2018) Global status report on alcohol and health 2018 Geneva
Wilson CRE, Gaffan D, Browning PGF, Baxter MG (2010) Functional localization within the prefrontal cortex: missing the forest for the trees? Trends Neurosci 33:533–540 https://doi.org/10.1016/j.tins.2010.08.001
Funding
LC-d-S was a recipient of the São Paulo Research Foundation—FAPESP scholarship (2016/08665–0). DB-d-S was a recipient of the Coordination for the Improvement of Higher Education Personnel—CAPES (88887.194785/2018–00) scholarship. RN-d-S (306556/2015–4) and CP (306557/2015–0) were recipients of the National Council for Scientific and Technological Development—CNPq research fellowship.
Fundação de Amparo à Pesquisa do Estado de São Paulo,2016/08665-0,Lucas Canto-de-Souza,Coordenação de Aperfeiçoamento de Pessoal de Nível Superior,88887.194785/2018-00,Daniela Baptista-de-Souza,Conselho Nacional de Desenvolvimento Científico e Tecnológico,306556/2015-4,Ricardo Luiz Nunes-de-Souza,306557/2015-0,Cleopatra Planeta
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethic approval
All experimental protocols were conducted according to the ethical principles of the Brazilian National Council for the Control of Animal Experimentation (CONCEA) and approved by the local Research Ethics Committee (CEUA/FCF-UNESP: 32/2016).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Canto-de-Souza, L., Baptista-de-Souza, D., Nunes-de-Souza, R.L. et al. Distinct roles of the left and right prelimbic cortices in the modulation of ethanol consumption in male mice under acute and chronic social defeat stress. Psychopharmacology 241, 1161–1176 (2024). https://doi.org/10.1007/s00213-024-06550-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-024-06550-8