Abstract
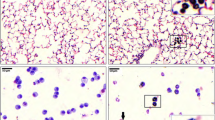
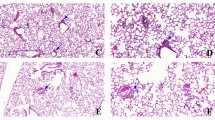
Arsenic trioxide (ATO), an inorganic arsenical, is a toxic environmental contaminant. It is also a widely used chemical with industrial and medicinal uses. Significant public health risk exists from its intentional or accidental exposure. The pulmonary pathology of acute high dose exposure is not well defined. We developed and characterized a murine model of a single inhaled exposure to ATO, which was evaluated 24 h post-exposure. ATO caused hypoxemia as demonstrated by arterial blood-gas measurements. ATO administration caused disruption of alveolar-capillary membrane as shown by increase in total protein and IgM in the bronchoalveolar lavage fluid (BALF) supernatant and an onset of pulmonary edema. BALF of ATO-exposed mice had increased HMGB1, a damage-associated molecular pattern (DAMP) molecule, and differential cell counts revealed increased neutrophils. BALF supernatant also showed an increase in protein levels of eotaxin/CCL-11 and MCP-3/CCL-7 and a reduction in IL-10, IL-19, IFN-γ, and IL-2. In the lung of ATO-exposed mice, increased protein levels of G-CSF, CXCL-5, and CCL-11 were noted. Increased mRNA levels of TNF-a, and CCL2 in ATO-challenged lungs further supported an inflammatory pathogenesis. Neutrophils were increased in the blood of ATO-exposed animals. Pulmonary function was also evaluated using flexiVent. Consistent with an acute lung injury phenotype, respiratory and lung elastance showed significant increase in ATO-exposed mice. PV loops showed a downward shift and a decrease in inspiratory capacity in the ATO mice. Flow-volume curves showed a decrease in FEV0.1 and FEF50. These results demonstrate that inhaled ATO leads to pulmonary damage and characteristic dysfunctions resembling ARDS in humans.







Similar content being viewed by others
Data availability
Data presented in this study are available from the corresponding author upon reasonable request.
References
Ahmad S, Zafar I, Mariappan N et al (2019) Acute pulmonary effects of aerosolized nicotine. Am J Physiol Lung Cell Mol Physiol 316(1):L94–L104. https://doi.org/10.1152/ajplung.00564.2017
Alimoghaddam K (2014) A review of arsenic trioxide and acute promyelocytic leukemia. Int J Hematol Oncol Stem Cell Res 8(3):44–54
Andersson U, Ottestad W, Tracey KJ (2020) Extracellular HMGB1: A therapeutic target in severe pulmonary inflammation including COVID-19? Mol Med 26(1):42. https://doi.org/10.1186/s10020-020-00172-4
Au WY (2011) A biography of arsenic and medicine in Hong Kong and China. Hong Kong Med J 17(6):507–513
Bolliger CT, van Zijl P, Louw JA (1992) Multiple organ failure with the adult respiratory distress syndrome in homicidal arsenic poisoning. Respir Int Rev Thor Dis 59(1):57–61. https://doi.org/10.1159/000196026
Chen R, Kang R, Tang D (2022) The mechanism of HMGB1 secretion and release. Exp Mol Med 54(2):91–102. https://doi.org/10.1038/s12276-022-00736-w
Egi M, Bellomo R, Stachowski E et al (2010) Hypoglycemia and outcome in critically ill patients. Mayo Clin Proc 85(3):217–224. https://doi.org/10.4065/mcp.2009.0394
Hassaballa HA, Lateef OB, Silver MR, Balk RA (2005) Acute lung injury induced by arsenic trioxide in a patient with refractory myelodysplastic syndrome. J Crit Care 20(1):111–113. https://doi.org/10.1016/j.jcrc.2004.08.009
Huaux F, Lasfargues G, Lauwerys R, Lison D (1995) Lung toxicity of hard metal particles and production of interleukin-1, tumor necrosis factor-alpha, fibronectin, and cystatin-c by lung phagocytes. Toxicol Appl Pharmacol 132(1):53–62. https://doi.org/10.1006/taap.1995.1086
Kahn T (1999) Hypernatremia with edema. Arch Intern Med 159(1):93–98. https://doi.org/10.1001/archinte.159.1.93
Krinsley JS, Keegan MT (2010) Hypoglycemia in the critically ill: how low is too low? Mayo Clin Proc 85(3):215–216. https://doi.org/10.4065/mcp.2010.0010
Kulkarni HS, Lee JS, Bastarache JA et al (2022) Update on the features and measurements of experimental acute lung injury in animals: an official american thoracic society workshop report. Am J Respir Cell Mol Biol 66(2):e1–e14. https://doi.org/10.1165/rcmb.2021-0531ST
Liu H, Liu Y, Shang Y, Liu H (2018) Toxicant deposition and transport in alveolus: a classical density functional prediction. Chem Res Toxicol 31(12):1398–1404. https://doi.org/10.1021/acs.chemrestox.8b00272
Manzoor S, Mariappan N, Zafar I et al (2020) Cutaneous lewisite exposure causes acute lung injury. Ann N Y Acad Sci 1479(1):210–222. https://doi.org/10.1111/nyas.14346
Mariappan N, Husain M, Zafar I et al (2020) Extracellular nucleic acid scavenging rescues rats from sulfur mustard analog-induced lung injury and mortality. Arch Toxicol 94(4):1321–1334. https://doi.org/10.1007/s00204-020-02699-1
Miller SI, Wallace RJ Jr, Musher DM, Septimus EJ, Kohl S, Baughn RE (1980) Hypoglycemia as a manifestation of sepsis. Am J Med 68(5):649–654. https://doi.org/10.1016/0002-9343(80)90250-8
Mingxing S, Haiying W, Congsong S, Chunyu Y, Liu C, Wang Q (2019) Acute toxicity of intratracheal arsenic trioxide instillation in rat lungs. J Appl Toxicol JAT 39(11):1578–1585. https://doi.org/10.1002/jat.3841
Muzaffar S, Khan J, Srivastava R, Gorbatyuk MS, Athar M (2022) Mechanistic understanding of the toxic effects of arsenic and warfare arsenicals on human health and environment. Cell Biol Toxicol. https://doi.org/10.1007/s10565-022-09710-8
Narendra DK, Hess DR, Sessler CN et al (2017) Update in management of severe hypoxemic respiratory failure. Chest 152(4):867–879. https://doi.org/10.1016/j.chest.2017.06.039
Neyra JA, Canepa-Escaro F, Li X et al (2015) Association of hyperchloremia with hospital mortality in critically Ill septic patients. Crit Care Med 43(9):1938–1944. https://doi.org/10.1097/CCM.0000000000001161
Palma-Lara I, Martinez-Castillo M, Quintana-Perez JC et al (2020) Arsenic exposure: a public health problem leading to several cancers. Regul Toxicol Pharmacol 110:104539 https://doi.org/10.1016/j.yrtph.2019.104539
Parvez F, Chen Y, Yunus M et al (2013) Arsenic exposure and impaired lung function. Findings from a large population-based prospective cohort study. Am J Respir Crit Care Med 188(7):813–819. https://doi.org/10.1164/rccm.201212-2282OC
Pinto SS, Enterline PE, Henderson V, Varner MO (1977) Mortality experience in relation to a measured arsenic trioxide exposure. Environ Health Perspect 19:127–130. https://doi.org/10.1289/ehp.7719127
Ratnaike RN (2003) Acute and chronic arsenic toxicity. Postgrad Med J 79(933):391–396. https://doi.org/10.1136/pmj.79.933.391
Rhoads K, Sanders CL (1985) Lung clearance, translocation, and acute toxicity of arsenic, beryllium, cadmium, cobalt, lead, selenium, vanadium, and ytterbium oxides following deposition in rat lung. Environ Res 36(2):359–378. https://doi.org/10.1016/0013-9351(85)90031-3
Sanchez TR, Powers M, Perzanowski M, George CM, Graziano JH, Navas-Acien A (2018) A meta-analysis of arsenic exposure and lung function: Is there evidence of restrictive or obstructive lung disease? Curr Environ Health Rep 5(2):244–254. https://doi.org/10.1007/s40572-018-0192-1
Shalaby KH, Gold LG, Schuessler TF, Martin JG, Robichaud A (2010) Combined forced oscillation and forced expiration measurements in mice for the assessment of airway hyperresponsiveness. Respir Res 11:82. https://doi.org/10.1186/1465-9921-11-82
Shebl E, Mirabile VS, Burns B (2022) Respiratory failure statpearls. Treasure Island (FL)
Solari R, Pease JE, Begg M (2015) Chemokine receptors as therapeutic targets: Why aren’t there more drugs? Eur J Pharmacol 746:363–367. https://doi.org/10.1016/j.ejphar.2014.06.060
Song K, Yang T, Gao W (2022) Association of hyperchloremia with all-cause mortality in patients admitted to the surgical intensive care unit: a retrospective cohort study. BMC Anesthesiol 22(1):14. https://doi.org/10.1186/s12871-021-01558-5
Srivastava RK, Li C, Ahmad A et al (2016) ATF4 regulates arsenic trioxide-mediated NADPH oxidase, ER-mitochondrial crosstalk and apoptosis. Arch Biochem Biophys 609:39–50. https://doi.org/10.1016/j.abb.2016.09.003
Stein EM, Tallman MS (2012) Provocative pearls in diagnosing and treating acute promyelocytic leukemia. Oncology (williston Park) 26(7):636–641
Villeda SA, Luo J, Mosher KI et al (2011) The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 477(7362):90–94. https://doi.org/10.1038/nature10357
Vriesendorp TM, DeVries JH, Hoekstra JB (2008) Hypoglycemia and strict glycemic control in critically ill patients. Curr Opin Crit Care 14(4):397–402. https://doi.org/10.1097/MCC.0b013e328306c7b1
Westervelt P, Brown RA, Adkins DR et al (2001) Sudden death among patients with acute promyelocytic leukemia treated with arsenic trioxide. Blood 98(2):266–271. https://doi.org/10.1182/blood.v98.2.266
Younan D, Richman J, Zaky A, Pittet JF (2019) An increasing neutrophil-to-lymphocyte ratio trajectory predicts organ failure in critically-ill male trauma patients. An exploratory study. Healthcare (Basel). https://doi.org/10.3390/healthcare7010042
Funding
Funding by the CounterACT Program grants, National Institutes of Health Office of the Director (NIH OD), the National Institute of Environmental Health Sciences (NIEHS) Grants U54ES030246 (MA, AfA, VA, AnA), U01ES025069 (AfA), U01ES028182 (SA), U01AR078544 (MA), U01ES033263 (SA), and R21ES030525 (SA) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
At the time of the study, AR was employed by SCIREQ Inc., a commercial entity with interests in topics related to the content of the present work. SCIREQ Inc. is an emka TECHNOLOGIES company. All the other authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mariappan, N., Zafar, I., Robichaud, A. et al. Pulmonary pathogenesis in a murine model of inhaled arsenical exposure. Arch Toxicol 97, 1847–1858 (2023). https://doi.org/10.1007/s00204-023-03503-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03503-6