Abstract
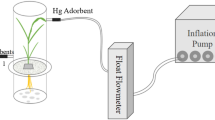
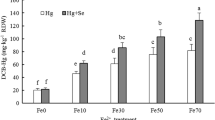
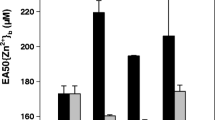
We investigated the ability of triticale uptake of Mercury (Hg), clarified whether triticale root uptake of Hg2+ via Zinc (Zn2+) transports, using hydroponic experiments. At 25℃, when Hg exposure in solution was lower than 20 μM, Hg concentration in the roots can be better described by a hyperbolic function, which shows a saturable characteristic. Under ice-cold (< 2℃) conditions, a nonsaturable (linear) component was found. Low exposure of Zn2+ (0–1 μM) inhibited plant Hg uptake when Hg exposure in the solution ranged from 1 to 10 μM, it showed an antagonistic effect of Zn on plant uptake of Hg. When Hg exposure was 20 μM, it revealed a synergistic effect of Zn on plant uptake of Hg, Hg in the root increased at the Zn (1 μM) exposure in the solution. Our results will deepen the understanding of Hg transfer in the soil–plant system.



Similar content being viewed by others
References
Chen J, Yang ZM (2012) Mercury toxicity, molecular response and tolerance in higher plants. Biometals 25(5):847–857. https://doi.org/10.1007/s10534-012-9560-8
Cohen CK (1998) The role of iron-deficiency stress responses in stimulating heavy-metal transport in plants. Plant Physiol 116(3):1063–1072. https://doi.org/10.1104/pp.116.3.1063
Daniels MJ, Mirkov TE, Chrispeels MJ (1994) The plasma membrane of Arabidopsis thaliana contains a mercury-insensitive aquaporin that is a homolog of the tonoplast water channel protein TIP. Plant Physiol 106(4):1325–1333. https://doi.org/10.1104/pp.106.4.1325
Eroglu K, Atli G, Canli M (2005) Effects of metal (Cd, Cu, Zn) interactions on the profiles of metallothionein-like proteins in the nile fish Oreochromis niloticus. Bull Environ Contam Toxicol 75(2):390–399. https://doi.org/10.1007/s00128-005-0766-0
Esteban E, Deza MJ, Zornoza P (2013) Kinetics of mercury uptake by oilseed rape and white lupin: influence of Mn and Cu. Acta Physiol Plant 35(7):2339–2344. https://doi.org/10.1007/s11738-013-1253-6
Feng X, Qiu G (2008) Mercury pollution in Guizhou, Southwestern China—an overview. Sci Total Environ 400(1–3):227–237. https://doi.org/10.1016/j.scitotenv.2008.05.040
Franciscato C, Moraes-Silva L, Duarte FA (2011) Delayed biochemical changes induced by mercury intoxication are prevented by zinc pre-exposure. Ecotoxicol Environ Saf 74(3):480–486. https://doi.org/10.1016/j.ecoenv.2010.11.011
Gupta M, Tripathi R, Rai U, Chandra P (1998) Role of glutathione and phytochelatin in Hydrilla verticillata (If) royle and Valusneria spiraus L. under mercury stress. Chemosphere 37(4):785–800. https://doi.org/10.1016/S0045-6535(98)00073-3
Haydon MJ, Cobbett CS (2010) Transporters of ligands for essential metal ions in plants. New Phytol 174(3):499–506. https://doi.org/10.1111/j.1469-8137.2007.02051.x
Hossain KFB, Hosokawa T, Saito T, Kurasaki M (2021) Zinc-pretreatment triggers glutathione and Nrf2-mediated protection against inorganic mercury-induced cytotoxicity and intrinsic apoptosis in PC12 cells. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2020.111320
Keltjens W, Van Beusichem M (1998) Phytochelatins as biomarkers for heavy metal stress in maize (Zea mays L.) and wheat (Triticum aestivum L.): combined effects of copper and cadmium. Plant Soil 203(1):119–126. https://doi.org/10.1023/A:1004373700581
Lane B, Kajioka R, Kennedy T (1987) The wheat-germ Ec protein is a zinc-containing metallothionein. Biochem Cell Biol 65(11):1001–1005. https://doi.org/10.1139/o87-131
Li P, Feng XB, Qiu GL, Shang LH, Li ZG (2009a) Mercury pollution in Asia: a review of the contaminated sites. J Hazard Mater 168(2–3):591–601. https://doi.org/10.1016/j.jhazmat.2009.03.031
Li Y, Yang L, Ji Y, Sun H, Wang W (2009b) Quantification and fractionation of mercury in soils from the Chatian mercury mining deposit, southwestern China. Environ Geochem Heal 31(6):617–628. https://doi.org/10.1007/s10653-008-9206-5
Li P, Du B, Chan HM, Feng X (2015) Human inorganic mercury exposure, renal effects and possible pathways in Wanshan mercury mining area, China. Environ Res 140:198–204. https://doi.org/10.1016/j.envres.2015.03.033
Liu F, Wang WX (2011) Differential roles of metallothionein-like proteins in cadmium uptake and elimination by the scallop Chlamys nobilis. Environ Toxicol Chem 30(3):738–746. https://doi.org/10.1002/etc.435
Liu F, Wang WX (2013) Facilitated bioaccumulation of cadmium and copper in the oyster Crassostrea hongkongensis solely exposed to zinc. Environ Sci Technol 47(3):1670–1677. https://doi.org/10.1021/es304198h
Liu N, Miao YJ, Zhou XX, Gan YD, Liu SW, Wang WX, Dai JL (2018) Roles of rhizos-pheric organic acids and microorganisms in mercury accumulation and translocation to different winter wheat cultivars. Agr Ecosyst Environ 258:104–112. https://doi.org/10.1016/j.agee.2018.02.005
Liu J, Wang J, Ning Y, Yang S, Wang P, Shaheen SM, Feng X, Rinklebe J (2019a) Methylmercury production in a paddy soil and its uptake by rice plants as affected by different geochemical mercury pools. Environ Int 129:461–469. https://doi.org/10.1016/j.envint.2019.04.068
Liu M, Zhang Q, Cheng M, He Y, Chen L, Zhang H, Cao H, Shen H, Zhang W, Tao S, Wang X (2019b) Rice life cycle-based global mercury biotransport and human methylmercury exposure. Nat Commun. https://doi.org/10.1038/s41467-019-13221-2
Mesquita M, Pedroso TF, Oliveira CS, Oliveira VA, do Santos RF, Bizzi CA, Pereira ME (2016) Effects of zinc against mercury toxicity in female rats 12 and 48 hours after H-gCl2 exposure. Excli J 15:256–267
Moons A (2003) Ospdr9, which encodes a PDR-type ABC transporter, is induced by heav-y metals, hypoxic stress and redox perturbations in rice roots. FEBS Lett 553(3):370–376. https://doi.org/10.1016/S0014-5793(03)01060-3
Moreno-Jimenez E, Penalosa JM, Esteban E, Carpena-Ruiz RO (2007) Mercury accumulation and resistance to mercury stress in Rumex induratus and Marrubium vulgare grown in perlite. J Plant Nutr Soil 170(4):485–494. https://doi.org/10.1002/jpln.200625238
Nowack B, Mayer K, Oswald S, Van Beinum W, Appelo C, Jacques D, Seuntjens P, Gérard F, Jaillard B, Schnepf A (2006) Verification and intercomparison of reactive transport codes to describe root-uptake. Plant Soil 285(1–2):305–321. https://doi.org/10.1007/s11104-006-9017-3
Obrist D, Kirk JL, Zhang L, Sunderland EM, Jiskra M, Selin NE (2018) A review of global environmental mercury processes in response to human and natural perturbations: changes of emissions, climate, and land use. Ambio 47(2):116–140. https://doi.org/10.1007/s13280-017-1004-9
Pandey P, Singh S (1993) Hg2+ uptake in a cyanobacterium. Curr Microbiol 26(3):155–159. https://doi.org/10.1007/BF01577371
Qiu G, Feng X, Jiang G (2012) Synthesis of current data for Hg in areas of geologic resource extraction contamination and aquatic systems in China. Sci Total Environ 421:59–72. https://doi.org/10.1016/j.scitotenv.2011.09.024
Rahman MM, Hossain KFB, Banik S, Sikder MT, Akter M, Bondad SEC, Rahaman MS, Hosokawa T, Saito T, Kurasaki M (2019) Selenium and zinc protections against metal-(loids)-induced toxicity and disease manifestations: a review. Ecotoxicol Environ Saf 168:146–163. https://doi.org/10.1016/j.ecoenv.2018.10.054
Rauser WE (1995) Phytochelatins and related peptides:structure, biosynthesis, and function. Plant Physiol 109:1141–1149. https://doi.org/10.1104/pp.109.4.1141
Rea PA (2007) Plant ATP-binding cassette transporters. Annual Rev Plant Biol 58(1):347–375. https://doi.org/10.1146/annurev.arplant.57.032905.105406
Rodríguez E, Peralta-Videa JR, Israr M, Sahi SV, Pelayo H, Sánchez-Salcido B, Gardea-Torresdey JL (2009) Effect of mercury and gold on growth, nutrient uptake, and anatomical changes in Chilopsis linearis. Environ Exp Bot 65(2):253–262. https://doi.org/10.1016/j.envexpbot.2008.09.014
Shi D, Wang W-X (2004) Understanding the differences in Cd and Zn bioaccumulation and subcellular storage among different populations of marine clams. Environ Sci Technol 38(2):449–456. https://doi.org/10.1021/es034801o
Smith RC, Epstein E (1964) Ion absorption by shoot tissue: kinetics of potassium and rubidium absorption by corn leaf tissue. Plant Physiol 39(6):992. https://doi.org/10.1104/pp.39.6.992
Sneller F, Van Heerwaarden L, Kraaijeveld-Smit F, Ten Bookum W, Koevoets P, Schat H, Verkleij J (1999) Toxicity of arsenate in Silene vulgaris, accumulation and degradation of arsenate-induced phytochelatins. New Phytol 144(2):223–232. https://doi.org/10.1046/j.1469-8137.1999.00512.x
Sun Q, Wang XR, Ding SM, Yuan XF (2005) Effects of interactions between cadmium and zinc on phytochelatin and glutathione production in wheat (Triticum aestivum L.). Environ Toxicol 20:195–201. https://doi.org/10.1002/tox.20095
Thangavel P, Long SR (2007) Changes in phytochelatins and their biosynthetic intermediates in red spruce (Picea rubens Sarg.) cell suspension cultures under cadmium and zinc stress. Plant Cell Tiss Org 88(2):201–216. https://doi.org/10.1007/s11240-006-9192-1
Walsh MJ, Goodnow SD, Vezeau GE, Richter LV, Ahner BA (2015) Cysteine enhances bioavailability of copper to marine phytoplankton. Environ Sci Technol 49(20):12145–12152. https://doi.org/10.1021/acs.est.5b02112
Wang JX, Feng XB, Anderson CWN, **ng Y, Shang LH (2012) Remediation of mercury contaminated sites—a review. J Hazard Mater 221:1–18. https://doi.org/10.1016/j.jhazmat.2012.04.035
Wang J, **a J, Feng X (2016) Screening of chelating ligands to enhance mercury accumulation from historically mercury-contaminated soils for phytoextraction. J Environ Manag. https://doi.org/10.1016/j.jenvman.2016.05.031
Wang J, Shaheen SM, Swertz A-C, Rennert T, Feng X, Rinklebe J (2019a) Sulfur-modified organoclay promotes plant uptake and affects geochemical fractionation of mercury in a polluted floodplain soil. J Hazard Mater 371:687–693. https://doi.org/10.1016/j.jhazmat.2019.03.010
Wang J, **ng Y, **e Y, Meng Y, **a J, Feng X (2019b) The use of calcium carbonate-enriched clay minerals and diammonium phosphate as novel immobilization agents for mercury remediation: spectral investigations and field applications. Sci Total Environ 646:1615–1623. https://doi.org/10.1016/j.scitotenv.2018.07.225
Xu J, Bravo AG, Lagerkvist A, Bertilsson S, Sjoblom R, Kumpiene J (2015) Sources and remediation techniques for mercury contaminated soil. Environ Int 74:42–53. https://doi.org/10.1016/j.envint.2014.09.007
Zhang X, Zhao FJ, Huang Q, Williams PN, Sun GX, Zhu YG (2009) Arsenic uptake and speciation in the rootless duckweed Wolffia globosa. New Phytol 182(2):421–428. https://doi.org/10.1111/j.1469-8137.2008.02758.x
Zhang H, Feng X, Larssen T, Qiu G, Vogt RD (2010) In Inland China, rice, rather than fish, is the major pathway for methylmercury exposure. Environ Health Perspect 118(9):1183–1188. https://doi.org/10.1289/ehp.1001915
Zhu DW, Zhong H, Zeng QL, Yin Y (2015) Prediction of methylmercury accumulation in rice grains by chemical extraction methods. Environ Pollut 199:1–9. https://doi.org/10.1016/j.envpol.2015.01.015
Zhu H, Zhong H, Wu J (2016) Incorporating rice residues into paddy soils affects methylmercury accumulation in rice. Chemosphere 152:259–264. https://doi.org/10.1016/j.chemosphere.2016.02.095
Acknowledgements
This study was funded by the Natural National Key R&D Program (No. 2017YFD0800302), the Natural Science Foundation of China (Nos. 4157312, 41703116, 41573082). The authors declare they have no financial interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, T., Man, Y., Li, P. et al. A Hydroponic Study on Effect of Zinc Against Mercury Uptake by Triticale: Kinetic Process and Accumulation. Bull Environ Contam Toxicol 108, 359–365 (2022). https://doi.org/10.1007/s00128-021-03298-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-021-03298-1