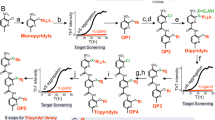
Abstract
In recent years, there has been growing interest in SARM1 as a potential breakthrough drug target for treating various pathologies of axon degeneration. SARM1-mediated axon degeneration relies on its TIR domain NADase activity, but recent structural data suggest that the non-catalytic ARM domain could also serve as a pharmacological site as it has an allosteric inhibitory function. Here, we screened for synthetic small molecules that inhibit SARM1, and tested a selected set of these compounds in a DRG axon degeneration assay. Using cryo-EM, we found that one of the newly discovered inhibitors, a calmidazolium designated TK106, not only stabilizes the previously reported inhibited conformation of the octamer, but also a meta-stable structure: a duplex of octamers (16 protomers), which we have now determined to 4.0 Å resolution. In the duplex, each ARM domain protomer is engaged in lateral interactions with neighboring protomers, and is further stabilized by contralateral contacts with the opposing octamer ring. Mutagenesis of the duplex contact sites leads to a moderate increase in SARM1 activation in cultured cells. Based on our data we propose that the duplex assembly constitutes an additional auto-inhibition mechanism that tightly prevents pre-mature activation and axon degeneration.






Similar content being viewed by others
Availability of data and material
All data and material will become available upon publication.
Change history
24 February 2023
author name Avraham Yaron was missed during proof stage and now it has been updated
23 February 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00018-023-04701-2
References
Osterloh JM, Yang J, Rooney TM, Fox AN, Adalbert R, Powell EH, Sheehan AE, Avery MA, Hackett R, Logan MA, MacDonald JM, Ziegenfuss JS, Milde S, Hou YJ, Nathan C, Ding A, Brown RH Jr, Conforti L, Coleman M, Tessier-Lavigne M, Zuchner S, Freeman MR (2012) dSarm/Sarm1 is required for activation of an injury-induced axon death pathway. Science 337:481–484. https://doi.org/10.1126/science.1223899
Gerdts J, Summers DW, Sasaki Y, DiAntonio A, Milbrandt J (2013) Sarm1-mediated axon degeneration requires both SAM and TIR interactions. J Neurosci 33:13569–13580. https://doi.org/10.1523/JNEUROSCI.1197-13.2013
Geisler S, Doan RA, Strickland A, Huang X, Milbrandt J, DiAntonio A (2016) Prevention of vincristine-induced peripheral neuropathy by genetic deletion of SARM1 in mice. Brain 139:3092–3108. https://doi.org/10.1093/brain/aww251
Cetinkaya-Fisgin A, Luan X, Reed N, Jeong YE, Oh BC, Hoke A (2020) Cisplatin induced neurotoxicity is mediated by Sarm1 and calpain activation. Sci Rep 10:21889. https://doi.org/10.1038/s41598-020-78896-w
Ozaki E, Gibbons L, Neto NG, Kenna P, Carty M, Humphries M, Humphries P, Campbell M, Monaghan M, Bowie A, Doyle SL (2020) SARM1 deficiency promotes rod and cone photoreceptor cell survival in a model of retinal degeneration. Life Sci Alliance. https://doi.org/10.26508/lsa.201900618
Ko KW, Milbrandt J, DiAntonio A (2020) SARM1 acts downstream of neuroinflammatory and necroptotic signaling to induce axon degeneration. J Cell Biol. https://doi.org/10.1083/jcb.201912047
Uccellini MB, Bardina SV, Sanchez-Aparicio MT, White KM, Hou YJ, Lim JK, Garcia-Sastre A (2020) Passenger mutations confound phenotypes of SARM1-deficient mice. Cell Rep 31:107498. https://doi.org/10.1016/j.celrep.2020.03.062
Kim Y, Zhou P, Qian L, Chuang JZ, Lee J, Li C, Iadecola C, Nathan C, Ding A (2007) MyD88-5 links mitochondria, microtubules, and JNK3 in neurons and regulates neuronal survival. J Exp Med 204:2063–2074. https://doi.org/10.1084/jem.20070868
Sasaki Y, Kakita H, Kubota S, Sene A, Lee TJ, Ban N, Dong Z, Lin JB, Boye SL, DiAntonio A, Boye SE, Apte RS, Milbrandt J (2020) SARM1 depletion rescues NMNAT1-dependent photoreceptor cell death and retinal degeneration. Elife. https://doi.org/10.7554/eLife.62027
Turkiew E, Falconer D, Reed N, Hoke A (2017) Deletion of Sarm1 gene is neuroprotective in two models of peripheral neuropathy. J Peripher Nerv Syst 22:162–171. https://doi.org/10.1111/jns.12219
Hughes RO, Bosanac T, Mao X, Engber TM, DiAntonio A, Milbrandt J, Devraj R, Krauss R (2021) Small molecule SARM1 inhibitors recapitulate the SARM1(-/-) phenotype and allow recovery of a metastable pool of axons fated to degenerate. Cell Rep 34:108588. https://doi.org/10.1016/j.celrep.2020.108588
Loring HS, Parelkar SS, Mondal S, Thompson PR (2020) Identification of the first noncompetitive SARM1 inhibitors. Bioorg Med Chem 28:115644. https://doi.org/10.1016/j.bmc.2020.115644
Bosanac T, Hughes RO, Engber T, Devraj R, Brearley A, Danker K, Young K, Kopatz J, Hermann M, Berthemy A, Boyce S, Bentley J, Krauss R (2021) Pharmacological SARM1 inhibition protects axon structure and function in paclitaxel-induced peripheral neuropathy. Brain. https://doi.org/10.1093/brain/awab184
Li WH, Huang K, Cai Y, Wang QW, Zhu WJ, Hou YN, Wang S, Cao S, Zhao ZY, **e XJ, Du Y, Lee CS, Lee HC, Zhang H, Zhao YJ (2021) Permeant fluorescent probes visualize the activation of SARM1 and uncover an anti-neurodegenerative drug candidate. Elife. https://doi.org/10.7554/eLife.67381
Shi Y, Kerry PS, Nanson JD, Bosanac T, Sasaki Y, Krauss R, Saikot FK, Adams SE, Mosaiab T, Masic V, Mao X, Rose F, Vasquez E, Furrer M, Cunnea K, Brearley A, Gu W, Luo Z, Brillault L, Landsberg MJ, Di Antonio A, Kobe B, Milbrandt J, Hughes RO, Ve T (2022) Structural basis of SARM1 activation, substrate recognition, and inhibition by small molecules. Mol Cell. https://doi.org/10.1016/j.molcel.2022.03.007
Bratkowski M, Burdett TC, Danao J, Wang X, Mathur P, Gu W, Beckstead JA, Talreja S, Yang YS, Danko G, Park JH, Walton M, Brown SP, Tegley CM, Joseph PRB, Reynolds CH, Sambashivan S (2022) Uncompetitive, adduct-forming SARM1 inhibitors are neuroprotective in preclinical models of nerve injury and disease. Neuron. https://doi.org/10.1016/j.neuron.2022.08.017
Essuman K, Summers DW, Sasaki Y, Mao X, DiAntonio A, Milbrandt J (2017) The SARM1 toll/interleukin-1 receptor domain possesses intrinsic NAD(+) cleavage activity that promotes pathological axonal degeneration. Neuron 93(1334–1343):e5. https://doi.org/10.1016/j.neuron.2017.02.022
Tong L (2021) How to diSARM the executioner of axon degeneration. Nat Struct Mol Biol 28:10–12. https://doi.org/10.1038/s41594-020-00545-7
Summers DW, Gibson DA, DiAntonio A, Milbrandt J (2016) SARM1-specific motifs in the TIR domain enable NAD+ loss and regulate injury-induced SARM1 activation. Proc Natl Acad Sci U S A 113:E6271–E6280. https://doi.org/10.1073/pnas.1601506113
Chuang CF, Bargmann CI (2005) A Toll-interleukin 1 repeat protein at the synapse specifies asymmetric odorant receptor expression via ASK1 MAPKKK signaling. Genes Dev 19:270–281. https://doi.org/10.1101/gad.1276505
Gerdts J, Brace EJ, Sasaki Y, DiAntonio A, Milbrandt J (2015) SARM1 activation triggers axon degeneration locally via NAD(+) destruction. Science 348:453–457. https://doi.org/10.1126/science.1258366
Horsefield S, Burdett H, Zhang X, Manik MK, Shi Y, Chen J, Qi T, Gilley J, Lai JS, Rank MX, Casey LW, Gu W, Ericsson DJ, Foley G, Hughes RO, Bosanac T, von Itzstein M, Rathjen JP, Nanson JD, Boden M, Dry IB, Williams SJ, Staskawicz BJ, Coleman MP, Ve T, Dodds PN, Kobe B (2019) NAD(+) cleavage activity by animal and plant TIR domains in cell death pathways. Science 365:793–799. https://doi.org/10.1126/science.aax1911
Sporny M, Guez-Haddad J, Lebendiker M, Ulisse V, Volf A, Mim C, Isupov MN, Opatowsky Y (2019) Structural evidence for an octameric ring arrangement of SARM1. J Mol Biol 431:3591–3605. https://doi.org/10.1016/j.jmb.2019.06.030
Sporny M, Guez-Haddad J, Khazma T, Yaron A, Dessau M, Shkolnisky Y, Mim C, Isupov MN, Zalk R, Hons M, Opatowsky Y (2020) Structural basis for SARM1 inhibition and activation under energetic stress. Elife. https://doi.org/10.7554/eLife.62021
Figley MD, Gu W, Nanson JD, Shi Y, Sasaki Y, Cunnea K, Malde AK, Jia X, Luo Z, Saikot FK, Mosaiab T, Masic V, Holt S, Hartley-Tassell L, McGuinness HY, Manik MK, Bosanac T, Landsberg MJ, Kerry PS, Mobli M, Hughes RO, Milbrandt J, Kobe B, DiAntonio A, Ve T (2021) SARM1 is a metabolic sensor activated by an increased NMN/NAD(+) ratio to trigger axon degeneration. Neuron 109(1118–1136):e11. https://doi.org/10.1016/j.neuron.2021.02.009
Jiang Y, Liu T, Lee CH, Chang Q, Yang J, Zhang Z (2020) The NAD(+)-mediated self-inhibition mechanism of pro-neurodegenerative Sarm1. Nature. https://doi.org/10.1038/s41586-020-2862-z
Bratkowski M, **e T, Thayer DA, Lad S, Mathur P, Yang YS, Danko G, Burdett TC, Danao J, Cantor A, Kozak JA, Brown SP, Bai X, Sambashivan S (2020) Structural and mechanistic regulation of the pro-degenerative NAD hydrolase SARM1. Cell Rep 32:107999. https://doi.org/10.1016/j.celrep.2020.107999
Shen C, Vohra M, Zhang P, Mao X, Figley MD, Zhu J, Sasaki Y, Wu H, DiAntonio A, Milbrandt J (2021) Multiple domain interfaces mediate SARM1 autoinhibition. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.2023151118
Bloom AJ, Mao X, Strickland A, Sasaki Y, Milbrandt J, DiAntonio A (2022) Constitutively active SARM1 variants that induce neuropathy are enriched in ALS patients. Mol Neurodegener 17:1. https://doi.org/10.1186/s13024-021-00511-x
Gilley J, Jackson O, Pipis M, Estiar MA, Al-Chalabi A, Danzi MC, van Eijk KR, Goutman SA, Harms MB, Houlden H, Iacoangeli A, Kaye J, Lima L, Queen Square G, Ravits J, Rouleau GA, Schule R, Xu J, Zuchner S, Cooper-Knock J, Gan-Or Z, Reilly MM, Coleman MP (2021) Enrichment of SARM1 alleles encoding variants with constitutively hyperactive NADase in patients with ALS and other motor nerve disorders. Elife. https://doi.org/10.7554/eLife.70905
Sasaki Y, Zhu J, Shi Y, Gu W, Kobe B, Ve T, DiAntonio A, Milbrandt J (2021) Nicotinic acid mononucleotide is an allosteric SARM1 inhibitor promoting axonal protection. Exp Neurol 345:113842. https://doi.org/10.1016/j.expneurol.2021.113842
Zhao ZY, **e XJ, Li WH, Liu J, Chen Z, Zhang B, Li T, Li SL, Lu JG, Zhang L, Zhang LH, Xu Z, Lee HC, Zhao YJ (2019) A Cell-Permeant Mimetic of NMN Activates SARM1 to Produce Cyclic ADP-Ribose and Induce Non-apoptotic Cell Death. iScience 15:452–466. https://doi.org/10.1016/j.isci.2019.05.001
Liu HW, Smith CB, Schmidt MS, Cambronne XA, Cohen MS, Migaud ME, Brenner C, Goodman RH (2018) Pharmacological bypass of NAD(+) salvage pathway protects neurons from chemotherapy-induced degeneration. Proc Natl Acad Sci U S A 115:10654–10659. https://doi.org/10.1073/pnas.1809392115
Loreto A, Angeletti C, Gu W, Osborne A, Nieuwenhuis B, Gilley J, Merlini E, Arthur-Farraj P, Amici A, Luo Z, Hartley-Tassell L, Ve T, Desrochers LM, Wang Q, Kobe B, Orsomando G, Coleman MP (2021) Neurotoxin-mediated potent activation of the axon degeneration regulator SARM1. Elife. https://doi.org/10.7554/eLife.72823
Wu T, Zhu J, Strickland A, Ko KW, Sasaki Y, Dingwall CB, Yamada Y, Figley MD, Mao X, Neiner A, Bloom AJ, DiAntonio A, Milbrandt J (2021) Neurotoxins subvert the allosteric activation mechanism of SARM1 to induce neuronal loss. Cell Rep 37:109872. https://doi.org/10.1016/j.celrep.2021.109872
Angeletti C, Amici A, Gilley J, Loreto A, Trapanotto AG, Antoniou C, Merlini E, Coleman MP and Orsomando G (2022) SARM1 is a multi-functional NAD(P)ase with prominent base exchange activity, all regulated bymultiple physiologically relevant NAD metabolites. iScience 25:103812. https://doi.org/10.1016/j.isci.2022.103812
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:42717. https://doi.org/10.1038/srep42717
Shacham-Silverberg V, Sar Shalom H, Goldner R, Golan-Vaishenker Y, Gurwicz N, Gokhman I, Yaron A (2018) Phosphatidylserine is a marker for axonal debris engulfment but its exposure can be decoupled from degeneration. Cell Death Dis 9:1116. https://doi.org/10.1038/s41419-018-1155-z
Cambronne XA, Stewart ML, Kim D, Jones-Brunette AM, Morgan RK, Farrens DL, Cohen MS, Goodman RH (2016) Biosensor reveals multiple sources for mitochondrial NAD(+). Science 352:1474–1477. https://doi.org/10.1126/science.aad5168
Houtkooper RH, Canto C, Wanders RJ, Auwerx J (2010) The secret life of NAD+: an old metabolite controlling new metabolic signaling pathways. Endocr Rev 31:194–223. https://doi.org/10.1210/er.2009-0026
Hara N, Osago H, Hiyoshi M, Kobayashi-Miura M, Tsuchiya M (2019) Quantitative analysis of the effects of nicotinamide phosphoribosyltransferase induction on the rates of NAD+ synthesis and breakdown in mammalian cells using stable isotope-labeling combined with mass spectrometry. PLoS ONE 14:e0214000. https://doi.org/10.1371/journal.pone.0214000
Liu L, Su X, Quinn WJ 3rd, Hui S, Krukenberg K, Frederick DW, Redpath P, Zhan L, Chellappa K, White E, Migaud M, Mitchison TJ, Baur JA, Rabinowitz JD (2018) Quantitative analysis of NAD synthesis-breakdown fluxes. Cell Metab. https://doi.org/10.1016/j.cmet.2018.03.018
Sasaki Y, Engber TM, Hughes RO, Figley MD, Wu T, Bosanac T, Devraj R, Milbrandt J, Krauss R, DiAntonio A (2020) cADPR is a gene dosage-sensitive biomarker of SARM1 activity in healthy, compromised, and degenerating axons. Exp Neurol 329:113252. https://doi.org/10.1016/j.expneurol.2020.113252
Ma S, Lapin D, Liu L, Sun Y, Song W, Zhang X, Logemann E, Yu D, Wang J, Jirschitzka J, Han Z, Schulze-Lefert P, Parker JE, Chai J (2020) Direct pathogen-induced assembly of an NLR immune receptor complex to form a holoenzyme. Science. https://doi.org/10.1126/science.abe3069
Martin R, Qi T, Zhang H, Liu F, King M, Toth C, Nogales E, Staskawicz BJ (2020) Structure of the activated ROQ1 resistosome directly recognizing the pathogen effector XopQ. Science. https://doi.org/10.1126/science.abd9993
Tal N, Morehouse BR, Millman A, Stokar-Avihail A, Avraham C, Fedorenko T, Yirmiya E, Herbst E, Brandis A, Mehlman T, Oppenheimer-Shaanan Y, Keszei AFA, Shao S, Amitai G, Kranzusch PJ, Sorek R (2021) Cyclic CMP and cyclic UMP mediate bacterial immunity against phages. Cell. https://doi.org/10.1016/j.cell.2021.09.031
Li X, Mooney P, Zheng S, Booth CR, Braunfeld MB, Gubbens S, Agard DA, Cheng Y (2013) Electron counting and beam-induced motion correction enable near-atomic-resolution single-particle cryo-EM. Nat Methods 10:584–590. https://doi.org/10.1038/nmeth.2472
Zhang K (2016) Gctf: Real-time CTF determination and correction. J Struct Biol 193:1–12. https://doi.org/10.1016/j.jsb.2015.11.003
Punjani A, Rubinstein JL, Fleet DJ, Brubaker MA (2017) cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat Methods 14:290. https://doi.org/10.1038/Nmeth.4169
Martinez M, Jimenez-Moreno A, Maluenda D, Ramirez-Aportela E, Melero R, Cuervo A, Conesa P, Del Cano L, Fonseca YC, Sanchez-Garcia R, Strelak D, Conesa JJ, Fernandez-Gimenez E, de Isidro F, Sorzano COS, Carazo JM, Marabini R (2020) Integration of Cryo-EM model building Software in scipion. J Chem Inf Model. https://doi.org/10.1021/acs.jcim.9b01032
Tegunov D, Cramer P (2019) Real-time cryo-electron microscopy data preprocessing with Warp. Nat Methods 16:1146–1152. https://doi.org/10.1038/s41592-019-0580-y
Winn MD, Ballard CC, Cowtan KD, Dodson EJ, Emsley P, Evans PR, Keegan RM, Krissinel EB, Leslie AG, McCoy A, McNicholas SJ, Murshudov GN, Pannu NS, Potterton EA, Powell HR, Read RJ, Vagin A, Wilson KS (2011) Overview of the CCP4 suite and current developments. Acta Cryst D 67:235–242. https://doi.org/10.1107/S0907444910045749
Krissinel E (2012) Enhanced fold recognition using efficient short fragment clustering. J Mol Biochem 1:76–85
Vagin A, Teplyakov A (2010) Molecular replacement with MOLREP. Acta Cryst D 66:22–25. https://doi.org/10.1107/S0907444909042589
Emsley P, Lohkamp B, Scott WG, Cowtan K (2010) Features and development of Coot. Acta Cryst D 66:486–501. https://doi.org/10.1107/S0907444910007493
Murshudov GN, Skubak P, Lebedev AA, Pannu NS, Steiner RA, Nicholls RA, Winn MD, Long F, Vagin AA (2011) REFMAC5 for the refinement of macromolecular crystal structures. Acta Cryst D 67:355–367. https://doi.org/10.1107/S0907444911001314
Lebedev AA, Young P, Isupov MN, Moroz OV, Vagin AA, Murshudov GN (2012) JLigand: a graphical tool for the CCP4 template-restraint library. Acta Crystallogr D Biol Crystallogr 68:431–440. https://doi.org/10.1107/S090744491200251X
Guez-Haddad J, Sporny M, Sasson Y, Gevorkyan-Airapetov L, Lahav-Mankovski N, Margulies D, Radzimanowski J, Opatowsky Y (2015) The neuronal migration factor srGAP2 achieves specificity in ligand binding through a two-component molecular mechanism. Structure 23:1989–2000. https://doi.org/10.1016/j.str.2015.08.009
Yom-Tov G, Barak R, Matalon O, Barda-Saad M, Guez-Haddad J, Opatowsky Y (2017) Robo Ig4 is a dimerization domain. J Mol Biol 429:3606–3616. https://doi.org/10.1016/j.jmb.2017.10.002
Demarest TG, Truong GTD, Lovett J, Mohanty JG, Mattison JA, Mattson MP, Ferrucci L, Bohr VA, Moaddel R (2019) Assessment of NAD(+)metabolism in human cell cultures, erythrocytes, cerebrospinal fluid and primate skeletal muscle. Anal Biochem 572:1–8. https://doi.org/10.1016/j.ab.2019.02.019
Maor-Nof M, Homma N, Raanan C, Nof A, Hirokawa N, Yaron A (2013) Axonal pruning is actively regulated by the microtubule-destabilizing protein kinesin superfamily protein 2A. Cell Rep 3:971–977. https://doi.org/10.1016/j.celrep.2013.03.005
Langer G, Cohen SX, Lamzin VS, Perrakis A (2008) Automated macromolecular model building for X-ray crystallography using ARP/wARP version 7. Nat Protoc 3:1171–1179. https://doi.org/10.1038/nprot.2008.91
Kandiah E, Giraud T, de Maria AA, Dobias F, Effantin G, Flot D, Hons M, Schoehn G, Susini J, Svensson O, Leonard GA, Mueller-Dieckmann C (2019) CM01: a facility for cryo-electron microscopy at the European Synchrotron. Acta Crystallogr D Struct Biol 75:528–535. https://doi.org/10.1107/S2059798319006880
Liebschner D, Afonine PV, Baker ML, Bunkoczi G, Chen VB, Croll TI, Hintze B, Hung LW, Jain S, McCoy AJ, Moriarty NW, Oeffner RD, Poon BK, Prisant MG, Read RJ, Richardson JS, Richardson DC, Sammito MD, Sobolev OV, Stockwell DH, Terwilliger TC, Urzhumtsev AG, Videau LL, Williams CJ, Adams PD (2019) Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr D Struct Biol 75:861–877. https://doi.org/10.1107/S2059798319011471
Afonine PV, Poon BK, Read RJ, Sobolev OV, Terwilliger TC, Urzhumtsev A, Adams PD (2018) Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr D Struct Biol 74:531–544. https://doi.org/10.1107/S2059798318006551
Acknowledgements
We acknowledge the European Synchrotron Radiation Facility for provision of beam time on CM01 and thank the staff of beamline CM01 of ESRF and members of the Opatowsky lab for technical assistance. We thank Gershon Kunin for IT management. The Israel National Center for Personalized Medicine is supported by a research grant from the Nancy and Stephen Grand. This work was supported by funds from ISF grants no. 1425/15 and 909/19, BSF grant no. 2019150, and ICRF grant 2022-2023 to Y.O. Y.O. is a Katzir Professorial Chair of Biophysics, and A.Y. is an incumbent of the Jack and Simon Djanogly Professorial Chair in Biochemistry.
Funding
This work was supported by funds from ISF grants no. 1425/15 and 909/19 and BSF grant no. 2019150 to Y.O.
Author information
Authors and Affiliations
Author notes
Tami Khazma and Yarden Golan-Vaishenker Co-first authors.
Yarden Opatowsky Lead contact.
- Yarden Opatowsky
Contributions
YO: Conceptualization, Resources, Formal analysis, Supervision, Funding acquisition, Validation, Investigation, Visualization, Methodology, Writing—original draft, Project administration, Writing—review and editing. Lead contact and corresponding author. TK: Data curation, Formal analysis, Investigation, Methodology. First author: YG-V: Formal analysis, Investigation, Methodology. Co-first author: JG-H: Data curation, Formal analysis, Supervision, Investigation, Methodology, Project administration, Writing—review and editing. AY: Contribution Conceptualization, Supervision, Methodology, Writing—review and editing. Co-corresponding author with YO. Atira Grossman: Data curation, Methodology, Investigation, Formal analysis, RS: Methodology. MW: Data curation, Methodology, AP: Data curation, Methodology, RZ: Contribution Data curation, Formal analysis, Methodology. MH: Data curation, Formal analysis, Validation, Methodology, Writing—review and editing. Co-corresponding author with YO.
Corresponding authors
Ethics declarations
Competing interests
We declare that we have no competing interests.
Consent for publication
We, all the authors give our consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
18_2022_4641_MOESM1_ESM.docx
Supplementary file1 (DOCX 498 KB) Figure 1 supplement 1. Structure-based sequence alignment of the SARM1 of human, mouse, zebrafish, and the C. elegans ortholog TIR-1. Color-coded highlights and Uniprot protein accession numbers are listed below.
18_2022_4641_MOESM2_ESM.docx
Supplementary file2 (DOCX 54 KB) Figure 3 supplement 1. A) Chemical structure of 2 additional hSARM1 inhibitory compounds. B) Determination of IC50 values, as in figure 3.
18_2022_4641_MOESM3_ESM.docx
Supplementary file3 (DOCX 130 KB) Figure 5 supplement 1. A) Representative cryo-EM micrograph of TK106 supplemented hSARM1. B) 2D class averages of the entire dataset. C) Flowchart of the cryo-EM processing steps of the entire dataset.
18_2022_4641_MOESM4_ESM.docx
Supplementary file4 (DOCX 196 KB) Figure 5 supplement 2. Resolution, angular distribution, and B-factor estimations of the cryo-EM maps of TK106 supplemented hSARM1 duplex and monoplex.
18_2022_4641_MOESM5_ESM.docx
Supplementary file5 (DOCX 174 KB) Figure 5 supplement 3. Comparison of available cryo-EM map densities of hSARM1, set to similar contour level at the ARM domain’s concave surface [24-27]. Dashed red oval marks the site of interest, highlighting the difference in map density between apo structures (empty site) and NAD+ or TK106 supplemented structures (occupied sites).
18_2022_4641_MOESM6_ESM.docx
Supplementary file6 (DOCX 177 KB) Figure 6 supplement 1. Extended representation of Fig. 6A. Protective and toxic effects of hSARM1 inhibitors over axon degeneration in mouse DRG explants.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khazma, T., Golan-Vaishenker, Y., Guez-Haddad, J. et al. A duplex structure of SARM1 octamers stabilized by a new inhibitor. Cell. Mol. Life Sci. 80, 16 (2023). https://doi.org/10.1007/s00018-022-04641-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04641-3