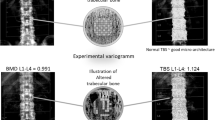
Abstract
The diagnosis of osteoporosis centers on assessment of bone mass and quality. In the absence of evidence-based guidelines to assess bone status in laboratory animals and unsuitability of use of T-/Z-scores meant for clinical application in animal studies, most investigators involved in new drug research and development employ clinical biomarkers and kits to assess bone turnover rate and portray change in bone mineral density (BMD) as percentage of increase/decrease, making comparative assessment of the effect highly impractical. This study proposes threshold boundaries of BMD (rT-score) in colony-bred Sprague-Dawley rats, distinct from those used clinically. Boundaries were obtained kee** fixed Type-I error (α=0.025). Femur neck was considered best for defining bone status using BMD measured by dual-energy X-ray absorptiometry. Findings demonstrate that BMD≤−1.96 rT-score signifies osteoporosis and BMD between >−1.96 and <−0.80 rT-score as osteopenia. Performance of boundaries to ascertain bone status was examined through simulation under different physiological/hormonal states viz. estrogen deficiency, ageing, estrus cycle, pregnancy, and lactation. The Area Under the Receiver Operating Characteristic curve of 0.98 obtained using BMD of femur neck, being close to unity, shows excellent ability of the proposed rT-score to effectively identify osteoporosis. Further studies using certain hierarchical measures of bone quality such as histomorphometry, mechanical testing etc. could supplement these findings. Since, unlike humans, most laboratory animals including rats only exhibit osteopenia and do not fracture their bones, the proposed thresholds are intended to serve as categorical tools to define bone quality and not to predict fracture risk.
Similar content being viewed by others
References
Kanis JA. Diagnosis of osteoporosis and assessment of fracture risk. Lancet 2002, 359: 1929–36.
Miller PD, Zapalowski C, Kulak CA, Bilezikian JP. Bone densitometry: the best way to detect osteoporosis and to monitor therapy. J Clin Endocrinol Metab 1999, 84: 1867–71.
WHO. Prevention and management of osteoporosis. WHO Technical Report series 921. 2000, 1–179.
Sengupta S, Arshad M, Sharma S, Dubey M, Singh MM. Attainment of peak bone mass and bone turnover rate in relation to estrus cycle, pregnancy and lactation in colony-bred Sprague-Dawley rats: suitability for studies on pathophysiology of bone and therapeutic measures for its management. J Steroid Biochem Molec Biol 2005, 94: 421–9.
Singh MM. Centchroman, a selective estrogen receptor modulator, as a contraceptive and for the management of hormone-related clinical disorders. Med Res Rev 2001, 21: 302–47.
Arshad M, Sengupta S, Sharma S, Ghosh R, Sawlani V, Singh MM. In vitro anti-resorptive activity and prevention of ovariectomy-induced osteoporosis in female Sprague-Dawley rats by ormeloxifene, a selective estrogen receptor modulator. J Steroid Biochem Molec Biol 2004, 91: 67–78.
Narayana Murthy PS, Sengupta S, Sharma S, Singh MM. Effect of ormeloxifene on ovariectomy-induced bone resorption, osteoclast differentiation and apoptosis and TGF beta-3 expression. J Steroid Biochem Mol Biol 2006, 100: 117–28.
Prummel MF, Wiersinga WM, Lips P, Sanders GT, Sauerwein HP. The course of biochemical parameters of bone turnover during treatment with corticosteroids. J Clin Endocrinol Metab 1991, 72: 382–6.
Alexandersen P, Riis BJ, Stakkestad JA, Delmas PD, Christiansen C. Efficacy of levormeloxifene in the prevention of postmenopausal bone loss and on the lipid profile compared to low dose hormone replacement therapy. J Clin Endocrinol Metab 2001, 86: 755–60.
The Area Under an ROC Curve. In: TG Tape. Interpreting Diagnostic Tests, http://gim.unmc.edu/dxtests/roc3.htm
Wronski TJ, Yen CF. The ovariectomized rat as an animal model for postmenopausal bone loss. Cells Mater 1991, 1(Suppl): 69–74.
Zeilmaker GH. Quantitative studies on the effect of the suckling stimulus on blastocyst implantation in the rat. Acta Endocrinol (Copenh) 1964, 46: 483–92.
Sowers MF, Holli BW, Shapiro B, et al. Elevated parathyroid hormone-related peptide associated with lactation and bone density loss. JAMA 1996, 276: 549–54.
Tommaselli GA, Guida M, Palomba S, Barbato M, Nappi C. Using complete breastfeeding and lactational amenorrhea as birth spacing methods. Contraception 2000, 61: 253–7.
INSA. Guidelines for Care and Use of Animals in Scientific Research. New Delhi: Indian National Science Academy 2000, 1–26.
Jimenez MA, Magee DE, Bryant HU, Turner RT. Clomiphene prevents cancellous bone loss from tibia of ovariectomized rats. Endocrinology 1997, 138: 1794–800.
Zarrow MX. Gestation. In: Young WC ed. Sex and Internal Secretions. 3rd ed. USA: Williams & Wilkins Co. 1967, 958–1031.
Weihe WH. Rattus norvegicus: The Laboratory Rat. In: Poole TB ed. The UFAW Handbook on the Care and Management of Laboratory Animals. Essex: Longman UK Ltd. 1967, 309–30.
Phillips AJ, Ostlere SJ, Smith R. Pregnancy-associated osteoporosis: does the skeleton recover? Osteoporosis Internat 2000, 11: 449–54.
Vajda EG, Bowman BM, Miller SC. Cancellous and cortical bone mechanical properties and tissue dynamics during pregnancy, lactation, and post-lactation in rat. Biol Reprod 2001, 65: 689–95.
Selby PL, Davies M, Adams JE. Do men and women fracture bones at similar bone densities? Osteoporos Int 2000, 11: 153–7.
Bagi CM, Wilkie D, Georgelos K, Williams D, Bertolini D. Morphological and structural correlates of the proximal femur in human and rat. Bone 1997, 21: 261–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, M., Mandal, S.K., Sengupta, S. et al. Quantification of bone mineral density to define osteoporosis in rat. J Endocrinol Invest 31, 393–399 (2008). https://doi.org/10.1007/BF03346382
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346382