Abstract
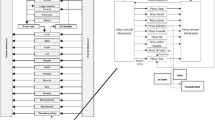
The disposition of pethidine (meperidine) in the pregnant rat is described by means of a physiological flow model. The model includes arterial and venous blood, brain, fat, fetal, hepatic, intestinal, muscular, pulmonar, and renal tissues. The concentration-time profiles of pethidine calculated by the model are consistent with experimental data, except for the brain and renal tissues, where the model predicts initially higher concentrations. Simulations are carried out to further explore the contribution from different organs on the kinetics in blood and tissues. The tissue-to-blood partition coefficients vary over a range from 5 to 316, where fat has the lowest and liver the highest after a correction is made due to hepatic extraction. Rapid uptake occurs into highly perfused organs such as brain, kidneys, liver, and lungs, followed by fetus, intestines, muscle, and fat. Data indicate no marked membrane resistance to pethidine of the investigated organs, except for fetal tissues, but rather a perfusion-limited uptake. Simulations suggest that muscles and adipose tissue play an important role in the rat, becoming the major reservoir of drug during the intermediate and terminal elimination phase, respectively. Volume of distribution and the biological half-life agree with reported findings. Pethidine is subject to a high systemic blood clearance, which exceeds the total hepatic blood flow in the rat. No degradation of pethidine is found in blood, and therefore a pulmonary expression for pethidine clearance is added as a potential source of pethidine elimination. The elimination of pethidine after a single i.v. bolus dose is found to be dependent on simulated changes in cardiac output and hepatic blood flow. A simulation is performed with the scaled model to mimic the human concentration-time profiles in maternal blood and brain tissues and fetal tissue during repetitive doses of pethidine.
Similar content being viewed by others
References
B. E. Dahlström, L. K. Paalzow, C. Lindberg, and C. Bogentoft. Pharmacokinetics and analgesic effect of pethidine (meperidine) and its metabolites in the ratDrug Metab. Dispos. 7:108–112 (1979).
L. E. Mather and P. J. Meffin. Clinical pharmacokinetics of pethidine.Clin. Pharmacokin. 3:352–368 (1978).
U. Klotz, T. S. McHorse, G. R. Wilkinson, and S. Schenker. The effect of cirrhosis on the disposition and elimination on meperidine in man.Clin. Pharmacol. Ther. 16:667–675 (1974).
D. Morgan, G. Moore, J. Thomas, and E. Triggs. Disposition of meperidine in pregnancy.Clin. Pharmacol. Ther. 23:288–295 (1977).
G. Lam, M.-L. Chen, and W. L. Chiou. Determination of tissue to blood partition coefficients in physiologically based pharmacokinetic studies.J. Pharm. Sci. 71:454–456 (1982).
J. V. Wait and F. Clarke III. DARE-P, A Portable Digital Simulation System, University of Arizona, Tucson, 1974.
H. Boxenbaum. Interspecies variation in liver weight, hepatic blood flow, and antipyrine clearance: Extrapolation of data to benzodiazepines and phenytoin.J. Pharmacokin. Biopharm. 8:165–175 (1981).
J. Buelke-Sam, J. F. Holson, and C. J. Nelson. Blood flow during pregnancy in the rat: II. Dynamics of and litter variability in uterine flow.Teratology 26:279–288 (1982).
E. D. Adolph. Quantitative relations in the physiological constitutions of mammals.Science 109:579–585 (1949).
W. L. Chiou. The physiological significance of total body clearance in pharmacokinetic studies.J. Clin. Hosp. Pharm. 7:25–30 (1982).
R. Bellman, J. A. Jacquez, and R. Kalaba. Some mathematical aspects of chemotherapy: I. Organ models.Bull. Math. Biophys. 22:181–198 (1960).
K. B. Bischoff and R. G. Brown. Drug distribution in mammals.Chem. Eng. Prog. Symp. Ser. 62:33–45 (1966).
K. B. Bischoff, R. L. Dedrick, and D. S. Zaharko. Preliminary model for methotrexate pharmacokinetics.J. Pharm. Sci. 59:149–154 (1970).
L. E. Gerlowski and R. K. Jain. Physiologically based pharmacokinetic modeling: Principles and applications.J. Pharm. Sci. 72:1103–1127 (1983).
G. E. R. Hook. The metabolic potential of the lungs. In C. F. George, D. G. Shand, and A. G. Renwick (eds.),Presystemic Drug Elimination. Butterworth, London, 1982, pp. 117–146.
S. L. Lindstedt and W. A. Calder. Body size, physiological time, and longevity of homeothermic animals.Q. Rev. Biol. 56:1–16 (1981).
G. K. Kramer, D. R. Gross, and C. Medlock. Contribution of the lung to total body clearance of meperidine in the dog.J. Pharm. Sci. 74:569–571 (1985).
L. M. Babiak, W. F. Cherry, S. Fayz, and K. S. Pang. Kinetics of meperidineN-demethyiation in the perfused rat liver preparation.Drug. Metab. Dispos. 12:698–704, (1984).
C. La Rosa, D. J. Morgan, and L. E. Mather. Pethidine binding in whole blood: Methodology and clinical significance.Br. J. Clin. Pharmacol. 17:405–409 (1984).
J. L. Gabrielsson and K. S. Larsson. The use of physiological pharmacokinetic models in studies on the disposition of salicylic acid in pregnancy. In H. Nau (ed.),Pharmacokinetics in Teratogenesis, CRC Press, Boca Raton, 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gabrielsson, J.L., Johansson, P., Bondesson, U. et al. Analysis of pethidine disposition in the pregnant rat by means of a physiological flow model. Journal of Pharmacokinetics and Biopharmaceutics 14, 381–395 (1986). https://doi.org/10.1007/BF01059198
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059198